2.2 Drug Regulation Process in Canada V2
There are many federal and provincial laws, as well as national guidelines, that have been established to protect public health and safety. This section will explain how Health Canada and provincial/territorial nursing governing bodies protect the public from medication harm.
Health Canada — Health Products and Food Branch
To protect the public, the Health Products and Food Branch (HPFB) of Health Canada is responsible for regulating, evaluating, and monitoring the safety, efficacy, and quality of therapeutic and diagnostic products available to Canadians. These products include drugs, medical devices, disinfectants, and sanitizers with disinfectant claims (Health Canada, 2020b). Some ways that the HPFB protects the public health regarding medications include:
- enforcing an official drug approval process based on evidence-based research;
- issuing safety warnings for medications with serious adverse reactions; and
- regulating over-the-counter (OTC) medications.
Each of these actions is further explained below.
Developing New Drugs
Canadian consumers benefit from having access to the safest and most advanced pharmaceutical system in the world. Drug companies conduct extensive research and work to develop and test a drug in a way that follows Health Canada’s Development and Approval Process of Drugs (SPharm Canada, n.d.).
Health Canada Approval — What it Means
Health Canada’s approval of a drug means that data on the drug’s effects have been reviewed by the HPFB, and the drug is determined to provide benefits that outweigh its known and potential risks for the intended population.
The HPFB reviews all new drug submissions and all the information about the drug captured during the development process (quality, preclinical, and clinical) and evaluates the risks of the drug versus its benefits to the Canadian population. More specifically, HPFB reviews information regarding the drug’s manufacturing, packaging, and labelling as well as information about the drug’s therapeutic claims and side effects. What doctors and clients will be told about the drug will also be reviewed, through the drug’s monographs and information sheets. All drugs allowed to be sold in Canada are reviewed to ensure that they meet the requirements of the Food and Drugs Act and its Regulations (Health Canada, 2025a). Once these requirements are met, the drug developer/sponsor receives a Notice of Compliance, confirming the dossier’s compliance with the Food and Drugs Act and its Regulations.
Safety Warnings
Clinical Reasoning and Decision-Making 1
Levofloxacin is an antibiotic that received FDA approval (the US equivalent of HFPB). However, after the drug was on the market, it was discovered that some clients who took levofloxacin developed serious, irreversible adverse effects, such as tendon rupture. The FDA issued a black box warning with recommendations to reserve levofloxacin for use in clients who have no alternative treatment options for certain indications, including uncomplicated UTI, acute exacerbation of chronic bronchitis, and acute bacterial sinusitis.
A nurse is preparing to administer medications to a client and notices that levofloxacin has been prescribed for the indication of pneumonia. There is no other documentation in the provider’s notes related to the use of this medication.
What is the nurse’s best response?
Note: Answers to the Clinical Reasoning activities can be found in the “Answer Key” sections at the end of the book.
Drug Schedules Regulation
Once a drug is approved, Health Canada will then determine if the drug will require a prescription for sale. Drugs that Health Canada has determined require a prescription for sale in Canada are listed on the Health Canada Prescription Drug List (PDL; Government of Canada, 2021) or in the schedules to the Controlled Drugs and Substances Act (1996) and its regulations (National Association of Pharmacy Regulatory Authorities, n.d.). From there, the role of National Association of Pharmacy Regulatory Authorities (NAPRA), the pharmacy regulatory body in Canada, is to regulate drugs at a provincial/territorial level and to ensure consistency across Canada. The Drug Schedules Regulation (1998) under the Pharmacy Operations and Drug Scheduling Act (2003) places all substances that are regulated under existing federal law into one of three schedules and four categories. The National Drug Schedule program also specifies further conditions of sale of any drug products.
Other products such as natural health products or medical devices are outside the scope of the National Drug Schedule program.
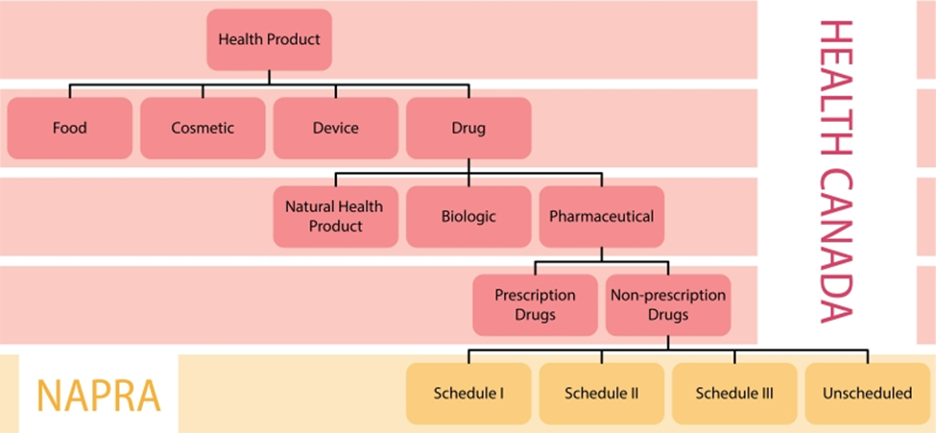
Figure 2,2a NAPRA drug schedules in Canada
https://www.napra.ca/national-drug-schedules/drug-scheduling-in-canada/
NAPRA was *Provided courtesy of the College of Pharmacy of Newfoundland and Labrador (cpnl.ca)
Permission needed
Health Professions and Occupations Act (HPOA)
Each province has its own health professions act that gives each healthcare profession’s regulatory body the ability to regulate or set the standards and scope of practice for their profession. In BC, the Health Professions Act has been replaced on April 1, 2026, with the Health Professions and Occupations Act. HPOA represents a significant modernization of the B.C.’s regulatory framework, with “an improved ability to protect the public, reinforce transparency and accountability and better reflects cultural safety, equity and the public interest” (BCCNM, 2026). As mentioned, HPOA establishes the legal framework for each health profession’s regulatory college. In BC, the British Columbia College of Nurses and Midwives (BCCNM) determine these standards of practice, as would College of Registered Nurses of Alberta in Alberta or the Nurse Association of New Brunswick.
In relation to medications, the regulatory college will determine the nurse’s authority to administer, dispense, and compound certain medications. Each nurse designation—nurse practitioner, registered nurse, licensed practical nurse, and registered psychiatric nurse—will have different regulations. It is important that nurses are aware of their standards of practice and regulations related to their licensing and regulation in the province in which they are working (BCCNM, 2025).
Scheduled Medications
The Drug Schedules Regulation (1998) under the Pharmacy Operations and Drug Scheduling Act (2003) places all substances regulated under existing federal law into one of three schedules or four categories. Scheduled medications are based on a substance’s medical use, potential for misuse, and safety or dependence liability.
The drug schedules are based on a cascading principle. A drug is first assessed against factors in Schedule 1. That drug will remain as a Schedule 1 drug if it meets the above criteria. If not, the drug is assessed against Schedule II factors, such as requiring professional intervention from a pharmacist at point of sale (professional services area). If not, then the drug is sold in the self-selection area of the pharmacy under direct supervision of a pharmacist (professional products area).
Because controlled substances have a greater chance of being misused, there are additional laws and procedures in place with these medications. A controlled substance, such as opioid medications, are placed in Schedule 1A.
Table 2.2.1 outlines each schedule, the type of drug that may be placed in that schedule, and a pharmacist and nurse’s responsibility in dispensing, compounding, and administering that drug.
| Schedule | Definition and RN Responsibility | Examples |
|---|---|---|
| Schedule I (Prescription) | Schedule I drugs require a prescription for sale and are provided to the public by a pharmacist following the diagnosis and professional intervention of a practitioner. The sale is controlled in a regulated environment as defined by provincial pharmacy legislation. |
|
| Schedule IA (Triplicate/Duplicate Prescription Program) | Drugs that may be sold by a pharmacist to a practitioner or on the prescription of a practitioner in accordance with Bylaw 5 (31) (6) of the bylaws to the Pharmacists, Pharmacy Operations and Drug Scheduling Act (2003).
This bylaw aims to reduce inappropriate prescribing of selected controlled drugs and to prevent forgeries. Prescriptions for the controlled drugs specified in the program must be written on the duplicate prescription pad specially developed for this purpose. |
|
| Schedule II (Professional Service Area) | Drugs that may be sold by a pharmacist on a non-prescription basis and that must be retained within the professional service area of the pharmacy where there is no public access and no opportunity for client self-selection. |
|
| Schedule III (Professional Products Area) | Drugs that may be sold by a pharmacist to any person from the self-selection professional products area of a licensed pharmacy. |
|
| Unscheduled (Non-pharmacy Sale) | Drugs that may be sold by a non-pharmacist to any person (e.g., convenience stores can carry these medications due to low risk to the public). |
|
For all nurses working in a facility, an order is required to administer medications. For nurses working in the community, they may be able to administer certain medications following a nursing diagnosis and using a decision support tool.
Example in Practice:
- A client has a minor ailment and needs a medication for treatment. In BC, pharmacists can prescribe medications for minor ailments such as urinary tract infections, allergies, acne, cold sores, contraceptives, and certain skin and fungal infections following an assessment and diagnosis. This recent change in policy utilizes a pharmacist’s full scope of practice.
- A nurse is working in a public health unit and will be giving vaccines in the flu clinic. The nurse can administer the vaccine following an assessment, nursing diagnosis, and using a decision support tool.
Acting Within Autonomous Scope of Practice (Without an Order)
In some circumstances, registered nurses (RNs) will be required to give certain medications without an order. They first need to do an assessment; based on a nursing diagnosis or using a decision support tool, they can give certain medications.
BCCNM — Acting Within Autonomous Scope of Practice
The Registered Nurse: Acting within Autonomous Scope of Practice standards, limits, and conditions apply to registered nurses and licensed graduate nurses when they are acting within autonomous scope of practice (without an order). These standards, limits, and conditions also apply to certified practice registered nurses when they are performing activities within their autonomous scope of practice (without an order) that are not within their certified practice designation.
Acting within autonomous scope of practice refers to registered nurses[1]:
- assuming accountability and responsibility for making decisions about client care, and
- performing activities that they are competent and allowed to perform without a client-specific order.
For the complete standard, go to BCCNM’s (n.d.a) Acting Within Autonomous Scope of Practice page. Note that standards will differ between provinces or countries. Nurses must always practice under the scope of practice for that jurisdiction.
Schedule I Medications
Under BCCNM’s (2025) standards of practice, RNs may prescribe, compound, dispense, or administer a limited number of Schedule I medications without an order and in an emergency. For example, in an emergency, RNs can give epinephrine for anaphylaxis or oxytocin for post-partum hemorrhage. As outlined below, nurses need to have additional education, complete an assessment and use a decision support tool prior to administration (BCCNM, 2025).
Treat:
- anaphylaxis
- cardiac dysrhythmia
- respiratory distress (clients with known asthma)
- hypoglycemia
- post-partum hemorrhage
Registered nurses may compound or administer:
- Epinephrine to treat anaphylaxis
- Epinephrine, atropine, amiodarone or lidocaine to treat cardiac dysrhythmia
- Salbutamol or ipratropium bromide to treat respiratory distress in known asthmatics
- Oral corticosteroids to treat respiratory distress in known asthmatics in emergency care settings
- D50W to treat hypoglycemia
- Oxytocin to treat post-partum hemorrhage
Registered nurses must follow a decision support tool if they:
- administer epinephrine to treat anaphylaxis.
- administer epinephrine, atropine, amiodarone or lidocaine to treat cardiac dysrhythmia and must also possess the competencies established by Providence Health Care.
- administer salbutamol, ipratropium bromide, or oral corticosteroids must also successfully complete additional education.
- administer D50W.
- administer oxytocin and must possess the competencies established by Perinatal Services BC (PSBC) and follow decision support tools established by PSBC (PSBC: Core Competencies for Management of Labour).
(BCCNM, 2025)
Schedule II Medications
In most cases, when nurses are treating a disease or disorder, they require a client-specific order. RNs can also compound, dispense, or administer Schedule II medications within autonomous scope of practice to treat a condition following an assessment and nursing diagnosis. This allows nurses to administer vaccines in health clinics, give glucagon for hypoglycemia, or give sublingual nitroglycerin for chest pain (BCCNM, 2025).
Drug Diversion
Drug diversion involves the transfer of any legally prescribed controlled substance from the individual for whom it was prescribed to another person for any illicit use. The most common drugs diverted from the healthcare facility setting are opioids. Tampering is the riskiest and most harmful type of diversion.
In some cases, drug diversion can occur by healthcare providers. Substance misuse by nurses is often unidentified, unreported, and untreated; nurses may continue to practise in places where their impairment may endanger the lives of their clients. BCCNM has established a professional and ethical responsibility to report a colleague’s suspected drug use. For more information about drug diversion in nurses visit BCCNM’s (n.d.b) Duty to Report: Narcotic Diversion and Substance Abuse Impairing Practice page.
Prescription Monitoring Programs (PMP)
In addition to drug diversion programs, prescription monitoring programs (PMP) have been established in several provinces to address prescription drug misuse, addiction, and diversion (Sproule, 2015). A PMP “collects information about prescription and dispensing of controlled substances for the purposes of monitoring, analysis, and education. In Canada, it is the responsibility of the provincial institutions to organize, maintain and run such programs” (Furlan et al., 2014). Providing valuable information about controlled substance prescriptions dispensed in the province aids healthcare professionals in their prescribing and dispensing decisions. The PMP also fosters the ability of pharmacies, healthcare professionals, law enforcement agencies, and public health officials to work together to reduce the misuse and diversion of prescribed controlled substance medications.
Proper Drug Disposal

Health Canada guidelines allow users to dispose of controlled substances in a safe and effective manner. A Johns Hopkins study on the sharing of medication found that:
- 60% of people had leftover opioids they hung on to for future use;
- 20% shared their medications;
- 8% would likely share with a friend;
- 14% would likely share with a relative; and
- only 10% securely locked their medication. (Open Resources for Nursing, 2023)
Health Canada also has a Take-back program in all provinces and territories that allows anyone to return unused medication at any time (see Figure 2.2.2). Additionally, needle disposal bins (yellow bins) are given through pharmacies for people on injectable medications.
Clinical Reasoning and Decision Making 2
- A nurse is providing discharge education to a client who recently had surgery and has been prescribed hydrocodone/acetaminophen tablets to take every four hours as needed at home. The nurse explains that when the post-op pain subsides and the medication is no longer needed, it should be dropped off at a local pharmacy for disposal in a collection receptacle. The client states, “I don’t like to throw anything away. I usually keep unused medication in case another family member needs it.”
What is the nurse’s best response?
- A nurse begins a new job on a medical-surgical unit. One of the charge nurses on this unit is highly regarded by her colleagues and appears to provide excellent care to her clients. The new nurse cares for a client that the charge nurse cared for on the previous shift. The new nurse asks the client about the effectiveness of the pain medication documented as provided by the charge nurse during the previous shift. The client states, “I didn’t receive any pain medication during the last shift.” The nurse mentions this incident to a preceptor who states, “I have noticed the same types of incidents have occurred with previous clients but didn’t want to say anything.
What is the nurse’s best response?
3. A nurse is working in a health clinic and is preparing to give a vaccine, a schedule 2 medication. The nurse can give the vaccine based on what actions were taken first?
Note: Answers to the Clinical Reasoning activities can be found in the “Answer Key” sections at the end of the book.
Learning Activities
References
British Columbia College of Nurses and Midwives. (n.d.a). Acting within autonomous scope of practice. https://www.bccnm.ca/RN/PracticeStandards/Pages/AutonomousSoP.aspx
British Columbia College of Nurses and Midwives. (n.d.b). Duty to report: narcotic diversion and substance use impairing practice. https://bccnm.ca/LPN/learning/dutytoreport/drug_diversion/Pages/reporting_responsibilities.aspx
British Columbia College of Nurses and Midwives (2026). Health Professions and Occupations Act takes effect in B.C. https://www.bccnm.ca/BCCNM/Announcements/Pages/Announcement.aspx?AnnouncementID=650
British Columbia College of Nurses and Midwives. (2025). Practice standard for all BCCNM nurses: Medication [PDF]. https://www.bccnm.ca/RN/PracticeStandards/Lists/GeneralResources/RN_PS_Medication.pdf
Controlled Drugs and Substances Act, S.C. c.19 (1996). https://laws-lois.justice.gc.ca/eng/acts/C-38.8/
Drug Schedules Regulation, BC Reg 9/98. https://www.bclaws.gov.bc.ca/civix/document/id/complete/statreg/9_98
Furlan, A. D., MacDougall, P., Pellerin, D., Shaw, K., Spitzig, D., Wilson, G., & Wright, J. (2014). Overview of four prescription monitoring/review programs in Canada. Pain Research & Management, 19(2), 102–106. https://doi.org/10.1155/2014/634171
Government of Canada. (2021). The drug and health product register. https://hpr-rps.hres.ca/pdl.php?lang=en
Health Canada. (2020a). Advisories, warnings and recalls – Drugs and health products. Government of Canada. https://www.canada.ca/en/health-canada/services/drugs-health-products/advisories-warnings-recalls.html
Health Canada. (2020b). How drugs are reviewed in Canada. Government of Canada. https://www.canada.ca/en/health-canada/services/drugs-health-products/drug-products/fact-sheets/drugs-reviewed-canada.html
Health Canada (2025a). Canada’s food and drug acts and regulations. Government of Canada. https://www.canada.ca/en/health-canada/services/food-nutrition/legislation-guidelines/acts-regulations/canada-food-drugs.html
Health Canada. (2025b). Canada vigilance adverse reaction online database. Government of Canada. https://www.canada.ca/en/health-canada/services/drugs-health-products/medeffect-canada/adverse-reaction-database.html
National Association of Pharmacy Regulatory Authorities. (n.d.). Drug scheduling in Canada. https://www.napra.ca/national-drug-schedules/drug-scheduling-in-canada/
Open Resources for Nursing. (2023). Nursing pharmacology (2nd ed.). Chippewa Valley Technical College. https://www.ncbi.nlm.nih.gov/books/NBK597872/
Pharmacy Operations and Drug Scheduling Act, S.B.C. c.77 (2003). https://www.bclaws.gov.bc.ca/civix/document/id/complete/statreg/03077_01
SPharm Canada. (n.d.). The drug review and approval process in Canada: An eGuide. https://spharm-inc.com/the-drug-review-process/?cn-reloaded=1
Sproule, B. (2015). Prescription monitoring programs in Canada: Best practice and program review [PDF]. Canadian Centre on Substance Abuse. https://campusmentalhealth.ca/wp-content/uploads/2018/03/CCSA-Prescription-Monitoring-Programs-in-Canada-Report-2015-en1.pdf
U.S. Department of Justice – Drug Enforcement Administration. (2017, December 13). Federal regulations and the disposal of controlled substances. https://www.deadiversion.usdoj.gov/mtgs/drug_chemical/2017/wingert.pdf#search=drug%20disposal
Media Attributions
- Figure 2.2a. NAPRA drug schedule
- Figure 2.2b Figure 2.2 Controlled Substances Collection Receptacle by Open Resources of Nursing, via Nursing Pharmacology (2nd ed.), is used under a CC BY 4.0 license.

