1.8 Pharmacology Principles — Lifespan Considerations V2
The safe use of medications requires healthcare providers to consider the unique pharmacokinetics and pharmacodynamics of drugs for different age groups and between the sexes. The nurse should consider the following questions:
- Are there special considerations related to the client’s age before I give this medication?
- Is the dose within acceptable limits?
- Do I need to check lab values before giving this medication?
- What is the client’s current status that may impact this medication?
- Does the client have any other health issues?
To begin this unit, we will examine the pharmacological differences between the sexes. The majority of this unit will examine lifespan considerations.
Sex Differences
Women have been historically underrepresented in drug trials and gender-specific drug analysis is still lacking, although there has been some improvement towards fair representation (Chu, 2014). Sex (referring to sex assigned at birth) differences are evident in body weight and plasma volume, along with all four phases of pharmacokinetics. These differences can contribute to less therapeutic effect and more adverse effects. Some of the differences are outlined below.
Absorption: Women have a slower GI transit time which affects the plasma concentration and absorption of oral drugs.
Distribution: In comparison to me, women have a higher percentage of body fat and lower body water content which can affect the volume of distribution of some drugs. For example, for lipophilic drugs like opioids, the drugs accumulate in body fat, and the half-life of lipophilic drugs in women is longer.
Metabolism: Differences in the metabolism of drugs with P450 enzymes makes metabolism phase the biggest difference between genders.
Excretion: Women have both a lower renal blood flow and glomerular filtration rate resulting in a slower clearance of drugs.
Although there is some differences with alterations in receptor number and receptor binding, pharmacodynamic differences are not as significant as pharmacokinetics.
For most drugs, gender-specific dosing recommendations are not considered. Women have more overdosages with meds, and more significant adverse effects. If a woman experiences more adverse effects from a drug or less therapeutic effect, a change in dosing may be necessary (Chu, 2014; Soldin & Mattison, 2013).
Pediatric Clients
In comparison to adults, children are not small adults. Whether the child is a newborn, a toddler, school aged, or a teen, there are pharmacokinetic differences that are considered by the practitioner when ordering medications. A pediatric client is much more sensitive to drugs than an adult and therefore much more at risk for adverse effects.
It is not just about the actual size difference of a pediatric client when comparing dosages between an infant and an adult, but size is considered. Up to the age of 12 years, most medications are based on weight or body surface area. Referring to the list below, many of these pharmacokinetic differences between adults and infants begin to diminish after the age of one, and continue to diminish until puberty. One exception are drugs that are metabolized faster in children. This requires alterations in dosing intervals or dosage amounts (Burcham & Rosenthal, 2019).
Did you know that lipophilic drugs have a higher distribution in infants than older children due to a higher body fat content—22% fat in a one year old vs 13% in a 15 year old.
Depending on the age, infants and young children will have:
- slower gastric emptying (slower peristalsis);
- reduced gastric acid secretion—increased bioavailability of acid-labile drugs (Penicillin) and decreased bioavailability of weakly acidic drugs (phenobarbital) (Tibbetts, 2020);
- more permeable intestine;
- body fat differences;
- higher body H2O;
- thinner skin—faster absorption of drugs;
- smaller muscle mass;
- Immature blood brain barrier (BBB)—increased permeability resulting in increased permeation of drugs;
- immature liver—decreased hepatic enzyme production and decreased first-pass elimination, resulting in higher drug levels in bloodstream; and
- immature kidneys—decreased excretion of a drug.
Consider how ADME will be impacted by each of these physiological differences. Infants and very young children are particularly vulnerable and are very sensitive to drugs.
Nursing Considerations for Pediatric Clients
All drug dosing must consider:
- age,
- body weight or body surface area (BSA, calculated by number of milligrams per metre squared (mg/m2)), and
- physiological changes related to illness (dehydration, sepsis, or other significant sickness can impact pediatric dosing for some medications).
Example in Practice
For any medication, always check for pediatric dosing. With the example below, the adult doses are the same regardless of age or weight, whereas the pediatric doses are based on the weight of the child.
Pediatric dosing for acetaminophen:
- PO (Adults and Children > 12 yrs): 325–650 mg every 6 hr, 1 g 3–4 times daily, or 1300 mg every 8 hr (not to exceed 3 g or 2 g/24 hr in patients with hepatic/renal impairments).
- PO (Children 1–12 yr): 10–15 mg/kg/dose every 6 hr as needed (not to exceed 5 doses/24 hr).
(Vallerand & Sanoski, 2023, p. 97).
Promoting Pediatric Medication Safety
Without question, administering a wrong dose to a child can result in client harm. Most adverse drug reactions (ADRs) are benign, but some can cause considerable harm and mortality. ADRs include medication errors such as a wrong dose, inappropriate use, or off-label use of a medication. A nurse working with pediatric clients needs to take additional training to ensure all procedures are followed. Some precautions that can be taken include:
- Liquid medications: Liquid medications are to be drawn up in a syringe or a med cup with measurements to ensure dosing accuracy. Spoons, teaspoons or droppers are inaccurate measuring devices and are subject to error. An oral syringe will not connect to a needleless port or accept a needle, and thus reducing the chance of a wrong route error (Smith, Leggett & Borg, 2022).
- High-risk medications: A nurse should adopt independent double checks for prescribing, preparing, and administering oral (e.g., liquid), intravenous (continuous and intermittent), or epidural medications for high-risk medications such as opioids, dopamine antagonists, and certain antibiotics among others (Smith, Leggett & Borg, 2022; Canadian Association of Pediatric Health Centers/Institute for Safe Medication Practices Canada, 2013).
Example in Practice:
Some medications are avoided in children; for example, the antibiotic Tetracycline is avoided because it stains developing teeth. Tetracycline should not be prescribed to children.
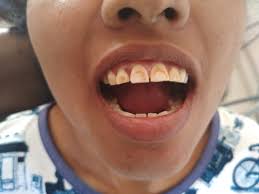
Figure 1.8a Dental flurosis (teeth with brown stains). https://commons.wikimedia.org/wiki/File:Dental_Flurosis_(teeth_with_brown_stains).jpg
Other medications are contraindicated. Acetylsalicylic acid (aspirin) is contraindicated in children due to risk of Reye’s Syndrome https://www.healthlinkbc.ca/healthwise/reye-syndrome
Pregnancy
Pregnancy poses challenges with medication use as most medications cross the placenta and are secreted in breast milk. Clinical trials on new or existing drugs ethically exclude pregnant women due to the concern for the impact on the fetus. As such, data on drug safety is limited and avoiding all medications is typically advised. There are times when avoiding medications during pregnancy is not an option. Previous chronic health issues, such as asthma, and pregnancy-induced conditions, such as hypertension and gestational diabetes, require treatment. Practitioners carefully consider initiating drugs during pregnancy, and the decision is made in collaboration with the client to review the risks and benefits to the health of the mother and the fetus.
Physiological Changes During Pregnancy
For pregnant women, the change in body composition can lead to changes in the plasma concentrations of a drug, which may result in too low of a concentration, leading to sub-therapeutic levels, or too high of one, leading to toxicity (World Health Organization [WHO], n.d.). Major physiological and anatomical changes occur in the cardiac system, liver, kidneys, and GI tract that can alter the pharmacokinetic and pharmacodynamics of the drugs administered. Depending on the trimester, delayed gastric emptying, blood volume and blood flow, renal filtration, hepatic enzymes (i.e., CYP3A, CYP2D6, CYP2C9), and transporters (P-GP) can be impacted (WHO, n.d.).
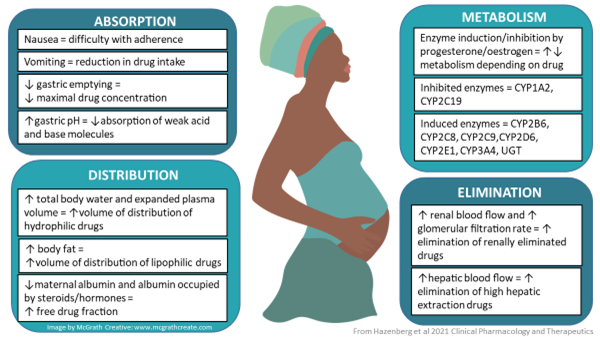
Andrea’s note: Ryu, R. & Hebert, M. (2022). Clinical Pharmacology during Pregnancy (2nd Ed.). Chapter 3 – impact of pregnancy on maternal pharmacokinetics of medications, 19-46. https://doi.org/10.1016/B978-0-12-818902-3.00015-4
Placental Drug Transfer
All drugs pass through the placenta and pass from the mother to the fetus by simple diffusion. Some drugs pass more readily than others, depending on the lipid solubility and molecular size of the drug, plasma drug levels in the mother, and blood flow to the placenta (Adams et al, 2018). Assume that all drugs taken during pregnancy will reach the fetus and, with that assumption, that all drugs need to be carefully considered.
Placenta
The placenta is a semi-permeable temporary membrane that slowly grows during the first 3 months of pregnancy. The placenta allows nutrition and gas exchange between the mother and fetus. This exchange is essential for the transfer of gases, electrolytes, hormones, maternal antibodies, fetal waste, and nutrition such as water, amino acids, glucose, vitamins, and free fatty acids (Herrick, E. & Bordoni, B., 2023). The placenta offers a degree of protection from maternal blood, filtering out some harmful substances, but almost all drugs cross the placenta.

Figure 1.8c Fetus and placenta. Fetus with umbilical cord, amniotic sac, and placenta. A cross section of the placenta shows how the fetal circulation interfaces with the maternal system without the mixing of blood.
File: Fetal circulation.jpg. File:Fetal circulation.jpg – Wikimedia Commons
Risk to Fetus
The first trimester (conception to 3 months) is the period of greatest risk to the developing fetus. This period is where skeletal and major organ development occurs and, as such, where developmental anomalies may occur. Birth defects can range from visible alterations, such as cleft palate, hydrocephalus or clubfoot, to metabolic and neurobehavioral effects (Burcham & Rosenthal, 2019). Other risks include low birth weight, premature labour, and spontaneous abortion. Less than 1% of birth defects are caused by drugs, with most birth defects due to genetic or environmental factors. If a pregnant client needs to take a drug for health reasons, if possible, a delay until after the first trimester is strongly considered.
During the second and third trimester, exposure to substances can still pose significant harm to the fetus. For example, during the third trimester the placenta becomes thinner, so there is a higher transfer of drugs from the maternal circulation to the fetus. The ability of the fetus to metabolize drugs is still underdeveloped, leading to a prolonged duration of that drug in the fetus.
A teratogen is any substance that can cause or increase the chance of a birth defect (Powell-Hamilton, 2025). Teratogen exposure may occur during pregnancy through exposure to radiation (i.e., X-rays) certain medications or drugs, or substance use. Fortunately, most pregnant clients who have been exposed to teratogens have newborns without birth defects; whether birth defects are present or not is dependent on the type of teratogen and length of exposure (Powell-Hamilton, 2025). The type of birth defect will depend on the fetal organ developing at that time.
Risk to Pregnant Client
With the physiological changes that occur during pregnancy, some drugs can have an adverse effect on the pregnant client. For example, the anticoagulant heparin can result in osteoporosis, which can eventually result in spinal compression fractures. Understanding the risks of any medications is important, and weighing the risks versus benefits is an important discussion with the healthcare provider.
Some health issues pose more risk to both the pregnant mother and fetus if left untreated, for example, pre-existing diabetes mellitus or gestational diabetes. Alternative medications can be prescribed, depending on what the client was on previously. For example, insulin is typically prescribed for gestational diabetes and is safe for the fetus with careful blood glucose control and monitoring. Other conditions such as heart failure can be treated with digoxin, which does not harm the fetus and will obviously benefit the expectant mother.
Unfortunately, some health conditions require the use of medications that may pose a risk to the fetus, or the pregnant client was exposed to a harmful drug prior to their knowledge of the pregnancy. Obtaining a drug history that includes exact dates of exposure and pregnancy dates is important to determine the risk.
Any client who is planning on becoming pregnant or is already pregnant should consult a healthcare provider. Nurses should obtain a detailed medication profile from the client that includes all prescribed medications, vitamins, over-the-counter medications, herbal supplements, and any recreational drug use. Nurses have an important role in ensuring clients are well-informed, seek the appropriate level of care, and are advocated for as needed.
Older Adult Clients
Pharmacokinetic Changes
Just like pediatric clients, older adults also needs special consideration. An older adult is defined as someone 65 years of age and older (Klotz, 2009). Changes in the body fat, water composition, and kidney and liver functioning will affect all stages of pharmacokinetics. As the client ages past middle adulthood, organ function progressively declines. Typically, the changes increase drug sensitivity due to declining renal and hepatic function. Significant individual variations can occur depending on the client’s level of fitness, lifestyle factors, and genetics.
Absorption: Although less affected by aging, older adults often have decreased gut motility and intestinal blood flow. There may be changes in the gastric (stomach) pH that may alter the absorption of certain medications. Their skin will be thinner leading to increased absorption of topical medications. Diminished fat stores can decrease the absorption of medications given transdermally.
Distribution: Older adults experience significant changes in body composition, as body fat increases and total body water and lean body mass decrease. The volume of distribution of polar drugs will decrease while lipophilic drugs will increase, requiring adjustments to drug dosages. Plasma protein binding and serum-albumin concentration changes are minimal in healthy older adults; however, if they are malnourished, it leads to a more pronounced decrease in albumin and protein binding sites. This will result in more free drugs in circulation and a higher risk of toxic drug levels.
Metabolism: With aging, there is approximately a 20–30% reduction in liver size and up to 50% reduction in hepatic blood flow. Hepatic enzymes may also diminish. With these changes, drug clearance may decrease, leading to prolonged half-lives and increased serum drug concentrations. There is significant variability with metabolism changes in older adults, with many experiencing minimal hepatic changes.
Elimination: Renal mass decreases along with a decrease in renal blood flow, which contributes to a decreased GFR. Always check renal function before giving new medications. GFR is the most accurate assessment of renal function as it is based on serum creatinine, sex, and age. Renal function is expected to decrease starting by age 40 as part of the normal aging process. By the age of 70, GFR may be reduced to 75 ml/hr/m2, signifying mild kidney impairment.
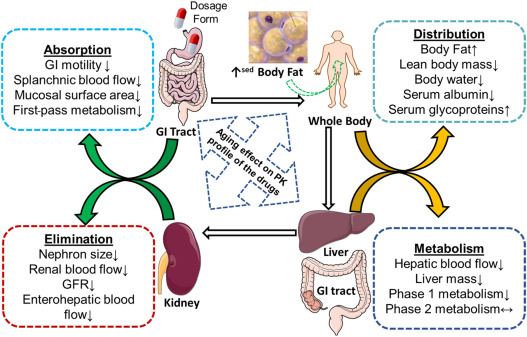
https://www.sciencedirect.com/science/article/abs/pii/B9780323983679000081
Please check if ok to use
Jessica / Nov 18, 2025: @Copyeditor please confirm if its ok to use image
Nursing Considerations For Older Adult Clients (> 65 Years of Age)
Nurses should consider the following factors when working with older adult clients:
- Check recent and past lab values (renal, hepatic, albumin)
- Start low and go slow
- Other:
- Weight—check for recent weight changes
- Body mass index (BMI)
- Thorough physical assessment including diet, bowel patterns, cardiac function, and SQ fat
- Always consider polypharmacy and drug-drug interactions
Medication Challenges With Older Adults
As a client ages, it is expected that some clients will develop health conditions that will need medications for treatment. As discussed earlier, the pharmacokinetic changes that occur in older adults make them more susceptible to medication issues. Some issues of concern are polypharmacy, medication non-adherence, and prescribing cascades that put the client more at risk of falls, frailty, or adverse drug effects. Refer to Chapter 3: Medication Safety: Preventing Medication Errors
References:
Bachelor, H. K., & Marriott, J. F. (2013). Pediatric pharmacokinetics: Key considerations. British Journal of Clinical Pharmacology, 79(3), 395–404. https://doi.org/10.1111/bcp.12267
Burcham, J. R., & Rosenthal, L. (2019). Lehne’s pharmacology for nursing care (10th ed.). Elsevier.
Canadian Association of Pediatric Health Centers, & Institute for Safe Medication Practices Canada. (2013). Canadian paediatric high alert medication delivery: Paediatric opioid safety – Phase 3: Education, knowledge translation and implementation [Final report, PDF]. https://www.ismp-canada.org/CurrentProjects/Paediatrics/downloads/20130117OpioidSafetyPhase3.pdf
Chippewa Valley Technical College, Egert, A., Lee, K., & Gill, M. (2023). Fundamentals of nursing pharmacology (1st Canadian ed.). BCcampus. https://opentextbc.ca/nursingpharmacology/chapter/1-9-examining-effect/
Chu, T. (2014). Gender differences in pharmacokinetics. US Pharmacist, 39(9):40-43.
Herrick, E. J., & Bordoni, B. (2023). Embryology, placenta. StatPearls. Retrieved October 20, 2025, from https://www.ncbi.nlm.nih.gov/books/NBK551634/
Klotz, U. (2009). Pharmacokinetics and drug metabolism in the elderly. Drug Metabolism Reviews, 41(2), 67–76.https://doi.org/10.1080/03602530902722679
Powell-Hamilton, N. N. (2025). Overview of birth defects. Merck Manual. Retrieved October 20, 2025, from https://www.merckmanuals.com/en-ca/home/children-s-health-issues/overview-of-birth-defects/overview-of-birth-defects
Robinson, P. (2021). Pharmacology, polypharmacy and the older adult: A review. British Journal of Community Nursing, 26(6), 290–295. https://doi.org/10.12968/bjcn.2021.26.6.290
Ryu, R., & Hebert, M. F. (2022). Chapter 3 – Impact of pregnancy on maternal pharmacokinetics of medications. In D. Mattison, & L-A. Halbert, Clinical pharmacology during pregnancy (2nd ed., pp. 19-46). https://doi.org/10.1016/B978-0-12-818902-3.00015-4
Sealock, K., & Seneviratne, C. (2021). Lilley’s pharmacology for Canadian health care professionals (4th ed.). Elsevier.
Smith, L., Leggett, C. & Borg, C. (2022). Administration of medicines to children: a practical guide. Australian Prescriber,45(6), 188-192. Administration of medicines to children: a practical guide – PMC doi: 10.18773/austprescr.2022.067
Soldin, O. & Mattison, D. (2013). Sex differences in pharmacokinetics and pharmacodynamics. Clinical Pharmacokinetics, 48(3):143-57. doi: 10.2165/00003088-200948030-00001. PMID: 19385708; PMCID: PMC3644551.
Tibbetts, E. (2025). Pharmacokinetics in children. Merck Manual. Retrieved Octob from https://www.Merckmanuals.com/en-ca/professional/pediatrics/prinicples-of-drug-treatment-in-children/pharmacokinetics-in-children
Vallerand, A. H., & Sanoski, C. A. (2023). Davis’s drug guide for nurses (18th ed.). F.A. Davis.
Varghese, D., Ishida, C., Patel, P., & Koya, H. H. (2024). Polypharmacy. StatPearls. Retrieved October 20, 2025, from https://www.ncbi.nlm.nih.gov/books/NBK532953/
Woods, D., Mentes, J., Cadogan, M. & Philips, L. (2017). Aging, genetic variations, and ethnopharmacology: Building cultural competence through awareness of drug responses in ethnic minority elders. Journal of Transcultural Nursing, 28(1), 56–62. https://doi.org/10.1177/1043659615606202
World Health Organization. (n.d.). Pharmacokinetics and dosing. Retrieved October 20, 2025, from https://www.who.int/tools/antiretrovirals-in-pregnancy-research-toolkit/pharmacokinetics-and-dosing
Media Attributions
- Figure 1.8a Dental flurosis (teeth with brown stains). https://commons.wikimedia.org/wiki/File:Dental_Flurosis_(teeth_with_brown_stains).jpg
- Figure 1.8b Pharmacokinetics and dosing during pregnancy
- File: Fetal circulation.jpg. File:Fetal circulation.jpg – Wikimedia CommonsFigure 1.8c
- Figure 1.8d “Figure 1: Changes in absorption, distribution, metabolism/biotransformation and excretion during pregnancy” by World Health Organization

