1.3 Pharmacokinetics — Absorption V2
Pharmacokinetics
Pharmacokinetics is the term that describes the four stages of absorption, distribution, metabolism, and excretion of drugs. Drugs are medications or other substances that have a physiological effect when introduced to the body.
There are four basic stages for a medication to go through within the human body:
- Absorption occurs after medications enter the body and travel from the site of administration into the body’s circulation.
- Distribution is the process by which medication is distributed throughout the body.
- Metabolism is the breakdown of a drug molecule.
- Excretion is the elimination of the medication from the body.
This entire process is sometimes abbreviated as ADME.
Absorption
The first stage of pharmacokinetics is known as absorption. Absorption is defined as the movement of the drug from its site of administration into the blood (Burcham & Rosenthal, 2019). Most drugs must be absorbed to produce an effect, with a few exceptions such as intestinal anti-infectives. The amount of absorption will determine how intense the effects will be, and the rate of absorption will determine how soon the effects will occur.
Drug absorption is impacted by a number of factors including the physical properties of the drug and the physiologic factors at the absorption site. For example, the formulation of the drug, either a tablet or liquid, determines the rate of absorption. An oral drug that is given in a tablet form must be dissolved before being absorbed, which delays absorption; whereas a liquid form of that drug does not need to dissolve will therefore have a faster absorption rate. Physiologic factors include the surface area (small intestine has a huge surface area compared to the stomach), blood flow (areas of high blood flow will absorb drugs more rapidly), lipid solubility, and pH levels. While most drugs need to be absorbed first before entering the bloodstream enroute to the target site, not all drugs do. For example, antacids are given orally and exert their actions locally in the stomach.
Absorption affects the speed and concentration at which a drug may arrive at its desired or target site. Each administration route has its own absorption characteristics, advantages, and disadvantages. The route chosen will depend on the desired effect and the type of drug preparations available. Depending on the intended response, the route is chosen based on the speed of response required (for example, the IV route is preferred for fast pain relief), duration of action, or other factors such as convenience, availability, and client factors.
Routes of Administration
Medications can enter the body through various routes of administration. The routes of administration are often divided into two major groups:
- Enteral, meaning through the gastrointestinal (GI) tract, includes all oral and rectal medications.
- Parenteral, literally meaning outside the GI tract but refers to by injection, includes intravenous, subcutaneous, intramuscular, intrathecally, and intra-articular medications.
Common routes to administer medications include:
- Oral (swallowing an aspirin tablet),
- Sublingual (dissolved under the tongue),
- Enteral (administering to the GI tract such as via a nasogastric tube),
- Rectal (administering a Tylenol suppository),
- Inhalation (breathing in medication from an inhaler),
- Intramuscular (getting a flu shot in the deltoid muscle),
- Subcutaneous (injecting insulin into the fat tissue beneath the skin), and
- Transdermal (wearing a nicotine patch).
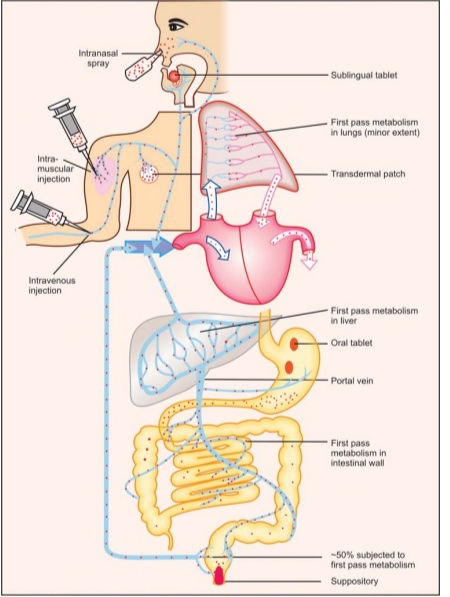
Characteristics of Routes of Administration
A new graduate nurse is planning on giving a client their medication and sees the prescriber orders have a few different routes for some of the medications. Which route should the nurse choose? What factors of the route, client, or intended effect would make one route more ideal than another?
Each route has advantages and challenges that can impact the effectiveness or are not as ideal for that client. The nurse uses the nursing process and clinical judgment in making the decision of which route to use when given the option. Depending on the orders, the nurse can decide to give a medication orally, IM, or IV. For example, the IV route requires the nurse to start an IV, which can be painful but IV meds have a rapid onset of action. In an emergency, rapid onset of action is highly important. If we need a more prolonged effect, IM might be the best option, but it can also be painful. If the client is able to tolerate an oral medication and its not an emergency, the oral route might be best due to its convenience.
Refer to the Table 1.3.2 for a review of route characteristics. The oral route will be examined more thoroughly below.
Oral Administration
The most common route of administration is the oral, or enteral, route. The drug is absorbed into the systemic circulation through the gastric mucosa, either in the stomach or small intestine. If given rectally, it is readily absorbed through the rectal mucosa.
Advantages of the oral route include convenience, affordable (compared to IV), and easy administration for most clients. The old saying, “if the gut works, use it,” would apply when choosing oral medications over IV ones. Other advantages are that the oral route has a high safety margin compared to IV routes, which could risk infection or fluid overload. It may also be possible to reverse an oral medication in the case of an overdose. If the medication was recently ingested, giving activated charcoal will bind with the medication to prevent absorption.

First-Pass Effect
First-pass metabolism is a major determinant of a drugs response or the peak plasma concentration of an oral dose. Medications made of protein, which are swallowed or otherwise absorbed in the stomach or small intestine, must then pass through the portal venous system and the liver sinusoids before entering the systemic circulation. The breakdown of the drug by the liver enzymes is known as the first-pass effect, and some of the drug will escape to the general circulation to either become protein bound (inactive) or stay free (and create an action at a receptor site).These metabolic effects are further described in the “Metabolism” section later in this chapter. Providers who prescribe medications, as well as nurses, understand that several doses of an oral medication may be needed before enough free drug stays active in the circulation to exert the desired effect.
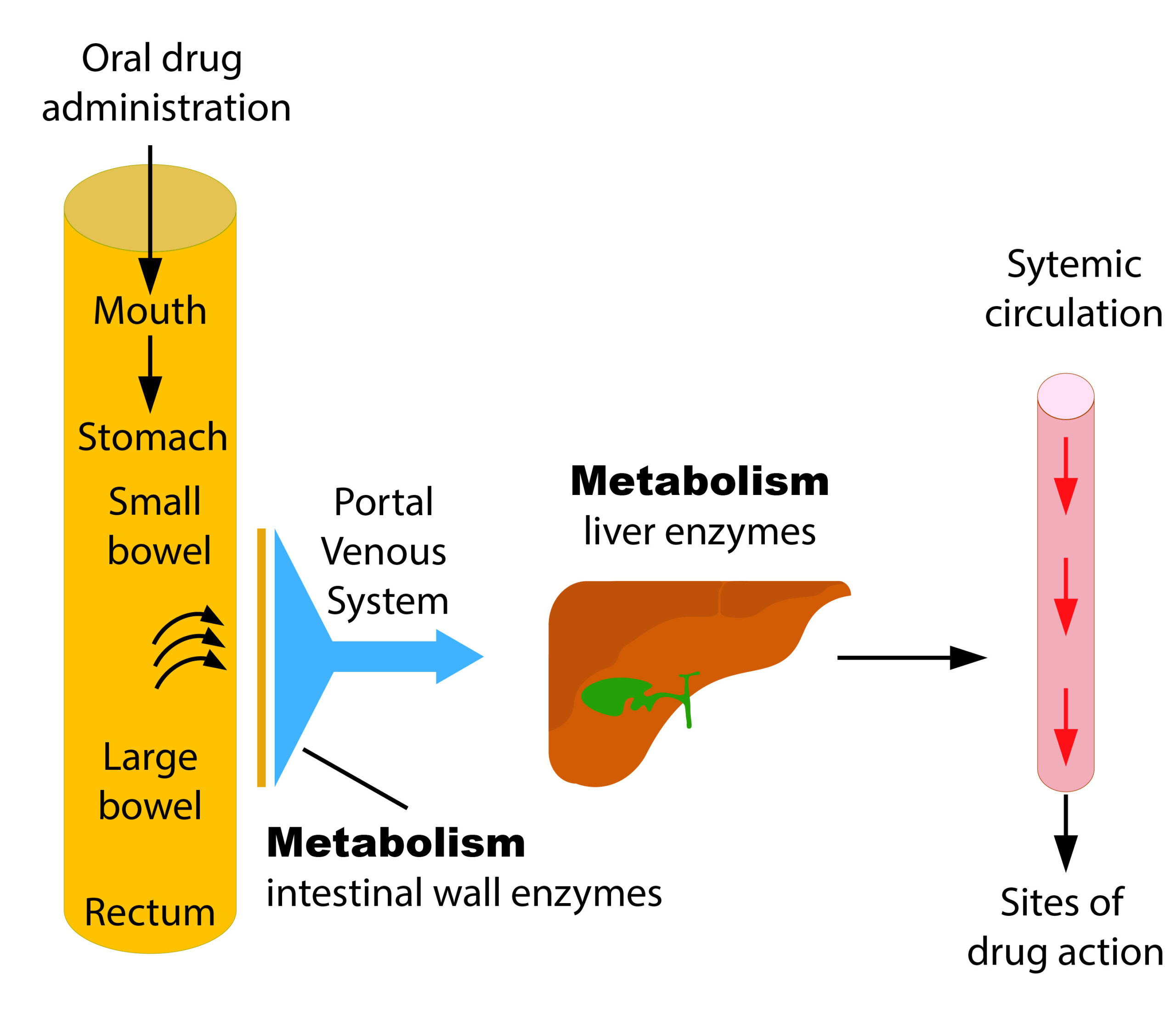
A workaround for the first-pass effect is to administer the medication using alternate routes, such as dermal, nasal, inhalation, subcutaneous, intramuscular, or intravenous. Alternative routes of medication administration bypass the first-pass effect by entering the bloodstream directly or via absorption through the skin or lungs. Intravenous medications administered directly into the bloodstream do not undergo absorption and are fully available for distribution to tissues within the body. Inhaling drugs through the nose or mouth is another alternative route for rapid medication delivery that bypasses the liver. For example, metered-dose inhalers have been a mainstay of asthma therapy for several years, and nasal steroid medications are often prescribed for allergy and sinus problems.
 Critical Thinking Out Loud
Critical Thinking Out Loud
A nurse reviews some new orders for a palliative care client. The order reads: Fentanyl 50 mcq/hr via transdermal patch.The nurse recalls some details and facts before preparing this medication:
- The client has had poor pain control with end stage pancreatic cancer. They are taking very little by mouth and do not want any more injections.
- Drugs delivered transdermally enter the blood via a meshwork of small arteries, veins, and capillaries in the skin. Transdermal drug delivery is particularly useful when medication must be administered over a long period of time to control symptoms.
- Rate of delivery of the active ingredient is stable and constant, and avoids first-pass effect.
- Transdermal patch is easy to apply and can stay on for 72 hours (typically) to 7 days.
- For application, check the orders and last time it was applied, remove the old patch, check for any forgotten patches, wear gloves, apply to intact skin, and do not apply any heat to the area (heating pad).
- Always ensure the 10 rights of medication administration are followed.
Transdermal application of medication is an alternate route that has the primary benefit of slow, steady drug delivery directly to the bloodstream—without passing through the liver first. (See Figure 1.3d for an image of applying a transdermal patch.)
Bioavailability
Bioavailability is the extent of drug absorption and is the fraction of the originally administered drug that arrives in systemic circulation. It plays a critical role in achieving the desired therapeutic effects. It is affected by many factors, including the properties of the drug, the mode of administration, interactions with other substances (food, fluids, other drugs), absorption (status and blood flow to small intestine), hepatic metabolism, and excretion (Stielow et al., 2023). For example, a client with gastric paresis post-operatively will slow the transit time through the gut.
A drug administered via the IV route, which bypasses the absorption phase, will have 100% of the drug arriving in the bloodstream. It is a fast delivery of the drug, and all of the drug dosage is delivered to the site, leading to 100% bioavailability. Orally administered drugs will have much lower bioavailability, as they must first go through the absorption phase and then be processed by digestive enzymes and the liver, which subsequently lowers the amount of drug that arrives in circulation. An example of a medication with low bioavailability is oral nitrates. Most routes have lower bioavailability than the gold standard of the IV route of administration. If two medications have the same bioavailability and also the same concentration of the active ingredient, then they are considered bioequivalent.
Two phrases are often heard when discussing bioavailability:
- Bioequivalent is if two medications given to the same client in the same dosage (concentration of active ingredient) result in equivalent concentrations of the drug in the plasma and tissues.
- Therapeutic equivalence is a step further and indicates that two medications given to the same client in the same dosage will have the same therapeutic and adverse effects.
It is expected that bioequivalent drugs are therapeutically equivalent. These terms are heard when comparing a generic drug with a brand-name drug.
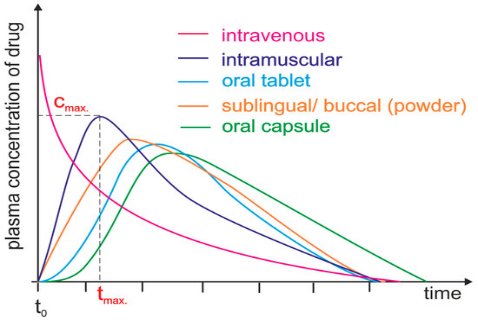
Delivering drugs directly into the systemic circulation via IV ensures 100% bioavailability. If they are delivered by SQ or IM, most drugs show between 60–100% bioavailability due to little or no metabolism in the skin or muscle. But the time to reach its maximum concentration is much longer than IV route.
If drugs are delivered by an oral route, bioavailability is lower, due to incomplete absorption and/or elimination during the first pass through the liver. Additionally, due to the indirect path to the plasma, they are characterized by a long-time lag (Stielow et al, 2023).

Many clients are prescribed a generic alternative to the brand-name drug, often due to the lower cost of the generic option. For example, a client’s drug plan will only cover the cheaper generic form. But is it the same? If its therapeutically equivalent, it should have the same therapeutic effects and adverse effects. If a client has more adverse effects with the generic version or does not get the same effectiveness as the brand-name product, this is called therapeutic nonequivalence. It is sometimes observed with clients who are on the brand-name drug for a long time and then switch to the generic form.
Oral Medication Preparations
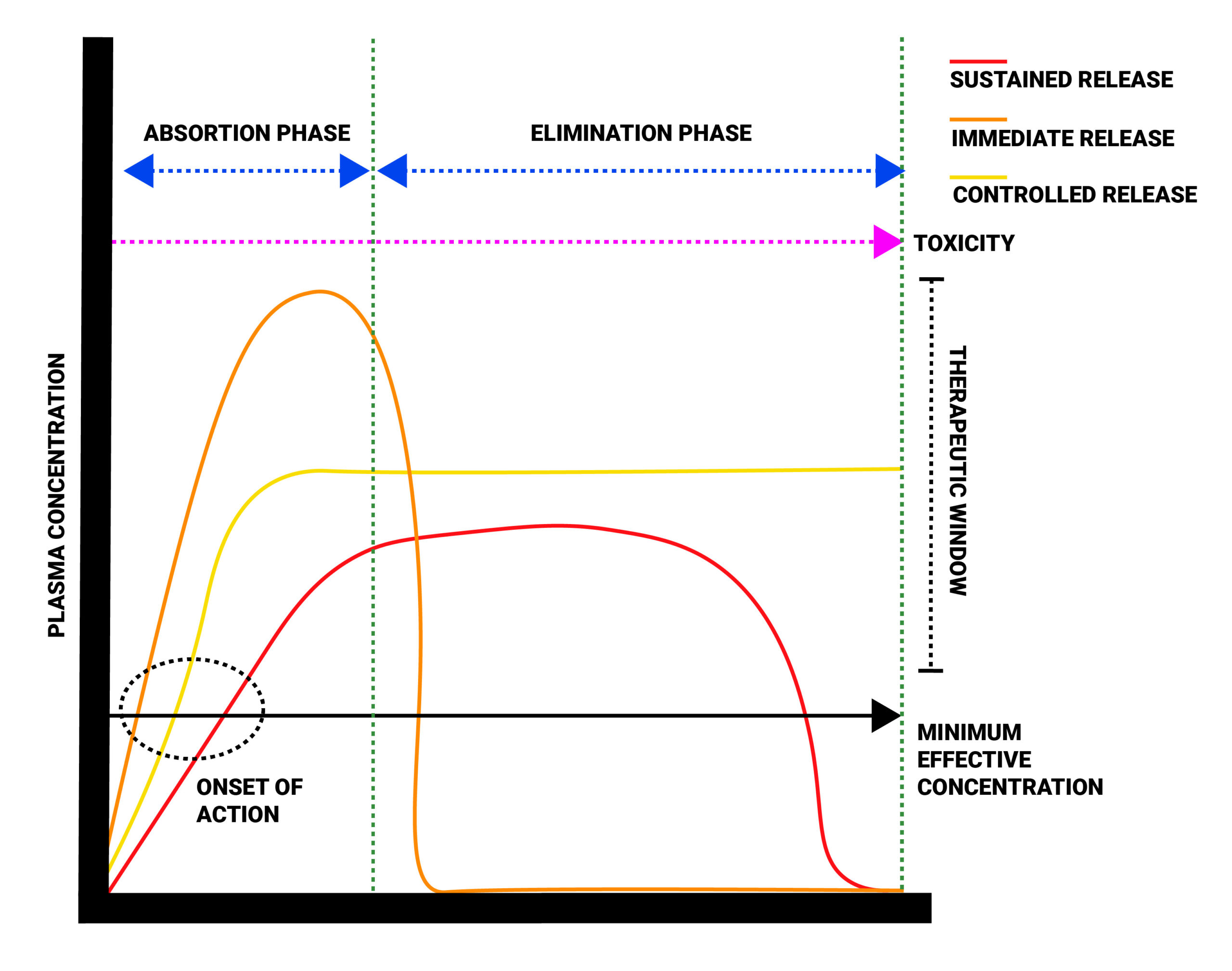
The route of the medication is not the only consideration when planning on giving an oral medication. The nurse must also check for how the med is ‘packaged’ or formulated. This applies to oral medication types like tablets or capsules. More specifically, they include enteric-coated, delayed-release, controlled-release, and sustained-release medications. Each type has different implications for the intensity of effect and duration of action (see Table 1.3.1), and impacts when the nurse will reassess for onset of action and monitoring for effects.
| Preparation Type | What Is It? | Benefits and Drawbacks |
|---|---|---|
| Enteric coated (EC) | The medication has been covered with a coating so it does not dissolve in the acidic environment of the stomach; instead, it dissolves in the alkaline environment of the intestine. | Benefit: Protects the stomach from drugs that may cause gastric irritation (e.g., Acetylsalicylic acid), or protects drugs from stomach acid.
Drawback: EC may not dissolve, leading to variable absorption rates. |
| Extended Release (ER) or (XR) | Two types: controlled and sustained release. These medications contain a larger amount of the drug that is designed to be released over a period of time. The capsule should not be opened or the tablet crushed to avoid releasing all the drug at once, altering the absorption rate and duration of action. | Benefits: Higher safety margin for narrow therapeutic window drugs. |
| Controlled release (CR) | Each capsule is designed to release the same amount of drug over a specified period of time. | Benefit: Controlled drug concentrations at the target site. |
| Sustained release (SR) | Each capsule is filled with tiny spheres of the drug. The coating of the spheres dissolve at variable rates, so some will dissolve immediately and others later in the day. | Benefit: Less doses during the day and a steadier state of the drug.
Drawback: More costly and some variable absorption rates. |
The Impact of Food on Absorption
Depending on the drug, you might see in the orders to give the medication with milk, without food, or even after a window of time where no food or drink can be taken. This is often ordered due to the impact on the absorption of that drug. Pay attention to these orders, for if they are not followed, clients may not be getting the ordered dose of that med or it can delay the onset of action leading to negative client outcomes.
There are many examples of drug-food interactions:
- Tetracycline, an antibiotic, cannot be given with food that contains calcium, iron, or magnesium, which can all delay absorption. It is best to take this antibiotic on an empty stomach.
- Levothyroxine, a thyroid medication, or lithium, a mood stabilizer, should not be taken with milk. Dairy products can bind with these drugs to make them ineffective. Ideally, take this medication on an empty stomach.
- Simvastatin, a cholesterol lowering medication, should not be taken with grapefruit juice. The juice will block with enzymes that metabolize the drug, leading to higher drug amounts and stronger effects. There are many meds that should not be taken with grapefruit. See the “Metabolism” unit for more details.
| Medication Route | Considerations |
|---|---|
Oral (PO), Sublingual (SL) or Enteral (NGT, GT, OGT) Ingestion
 |
Oral route (PO), Enteral
Sublingual/Buccal:
|
Rectal (PR)
 |
Rectal:
|
Parenteral Injection (SC, IM, IV)
 |
Subcutaneous (SQ) and intramuscular (IM) administration:
SQ:
IM:
SQ and IM:
Intravenous (IV):
|
Pulmonary Inhalation
 |
Inhalation:
|
Topical and Transdermal Application
 |
Topical creams, lotions, and ointments:
Transdermal patch:
|
Researchers who specialize in pharmacokinetics must also pay attention to another dimension of drug action within the body: time. Unfortunately, scientists do not have the ability to actually see where a drug is going or how long it is active. To compensate, they use mathematical models and precise measurements of blood and urine to determine where a drug goes and how much of the drug (or breakdown product) remains after the body processes it. Other indicators, such as blood levels of liver enzymes, can help predict how much of a drug is going to be absorbed.
Principles of chemistry are also applied while studying pharmacokinetics because the interactions between drugs and body molecules are really just a series of chemical reactions. Understanding the chemical encounters between drugs and biological environments, such as the bloodstream and the oily surfaces of cells, is necessary to predict how much of a drug will be metabolized by the body.
Learning Activities
Clinical Reasoning and Decision-Making Activities
- Choosing between two routes of administration: A nurse is planning on giving acetaminophen for a client experiencing moderate pain. The order reads:
- Acetaminophen 325-650 mg PO q 4-6 hours prn
- Acetaminophen 325-650 mg PR q 4-6 hours prn
Applying the principles of absorption for both routes, which factors should the nurse consider when considering which route to give?
- A client is in acute 9/10 pain. The nurse wants to give the Morphine IV as opposed to IM. What is the rationale to support this decision?
Note: Answers to the Clinical Reasoning activities can be found in the “Answer Key” sections at the end of the book.
References
Burcham, J. R., & Rosenthal, L. (2019). Lehne’s pharmacology for nursing care (10th ed). Elsevier.
Doyle, G. R., & McCutcheon, J. A. (2015). 6.7 Administering topical medication. In Clinical procedures for safer patient care. BCcampus. https://opentextbc.ca/clinicalskills/chapter/administering-topical-medication/. Download this book for free at http://open.bccampus.ca.
Polaka, S., Tella., J. D., Tekade, M., Sharma, M., & Tekade, R. K. (2022). Chapter 10 – Impact of ageing on pharmacokinetics and pharmacodynamics of the drugs. In R. K. Tekade (Ed.), Pharmacokinetics and toxicokinetic considerations (p. 241-261). Academic Press. https://doi.org/10.1016/B978-0-323-98367-9.00008-1
Stielow, M., Witczyńska, A., Kubryń, N., Fijałkowski, Ł., Nowaczyk, J., & Nowaczyk, A. (2023). The bioavailability of drugs—The current state of knowledge. Molecules, 28(24), Article 8038. https://doi.org/10.3390/molecules28248038
Media Attributions
- Symbol lightbulb by CheChe, via Wikimedia Commons, is used under a CC BY-SA 4.0 license.
- Generic Brands Same Quality and Performance by U.S. Food and Drug Administration, in the public domain.
- Figure 1.3a A drug’s life in the body (with labels) | Medicines taken by NIH Image Gallery, via Flickr, is used under a CC BY-NC 2.0 license.
- Figure 1.3b Person Holding White Medication Pills by Ahsanjaya, via Pexels, is used under the Pexels License.
- Figure 1.3c Applying Transdermal Patch by Glynda Rees Doyle and Jodie Anita McCutcheon (2015), via BCIT’s Clinical Procedures for Safer Patient Care, is used under a CC BY 4.0 license.
- Figure 1.3d Plasma level time curves for different types of drug administration is from Stielow et al. (2023), and is used under the MDPI Open Access terms.
- Figure 1.3e A comparison of brand name and generic medications is by Sheila Odubote and is subject to the CC BY NC SA license.
- Figure 1.3f Plasma concentration over time for different drug-release formulations is by TRU Open Press and is subject to the CC BY NC SA license.
- Table 1.3g:
- Figure 1.3h Administering oral medication by Glynda Rees Doyle and Jodie Anita McCutcheon (2015), via BCIT’s Clinical Procedures for Safer Patient Care, is used under the CC BY 4.0 license.
- Figure 1.3i Administering-med-rectally-2 [Figure 6.1] by Glynda Rees Doyle and Jodie Anita McCutcheon (2015), via BCIT’s Clinical Procedures for Safer Patient Care, is used under the CC BY 4.0 license.
- Figure 1.3j Subcutaneous (SQ) and intramuscular (IM) administration by National Cancer Institute, via Unsplash, is used under the Unsplash License.
- Figure 1.3k Adult Using an Asthma Inhaler (29251369035) by National Institute of Allergy and Infectious Diseases (NIAID), via Flickr, is used under a CC BY 2.0 license.
- Figure 1.3l Applying Transdermal Patch by Glynda Rees Doyle and Jodie Anita McCutcheon (2015), BCIT’s Clinical Procedures for Safer Patient Care, is used under a CC BY 4.0 license.
The study of how the body absorbs, distributes, metabolizes, and eliminates drugs.
Medications or other substances that have a physiological effect when introduced to the body.
The first stage of pharmacokinetics: medications enter the body and travel from the site of administration into the body's circulation.
The second stage of pharmacokinetics; the process by which medication is distributed throughout the body.
The breakdown of a drug molecule via enzymes in the liver (primarily) or intestines (secondarily).
The final stage of pharmacokinetics; process by which the body eliminates waste or excess.

