3.7 Medication Safety in High-Risk Situations
Most nurses have worked in a situation where, despite their best intentions, the risk of medication errors is high.
Example in practice: A medical unit nurse with three years of experience has been floated to the emergency department as they are short staffed. The nurse is given six ‘stable’ ED patients but has more clients than the allowed ratio. The ages range from a 6 month old with suspected pneumonia to a 40 year old with stable angina to a 90 year old with a fractured hip and history of dementia. The nurse is given a quick orientation to the unit and then proceeds to assess her clients.
Does this scenario sound familiar? Can you identify potential risks to client safety? All these clients will be prescribed medications. What challenges can you see with the nurse administering the meds?
From the above example, it is easy to identify many areas where things could go wrong and pose a risk to the client. The following unit will examine categories to improve medication safety, including high-risk medications, healthcare provider, systems, and client factors. Note that each strategy can also be applied more broadly to other categories.
High-Risk (High-Alert) Medications
High-risk medications are drugs that bear a heightened risk of causing significant client harm when they are used in error. Medication incidents can include harmful events, no-harm events, near misses, and circumstances that can lead to harmful incidents. The Institute for Safe Medication Practices (ISMP) Canada (2020) prepared a report highlighting the degree of harm with medication in specific practice settings using data from ISMP’s Individual Practitioner Reporting, Consumer Reporting, and National Incident Data Repository for Community Pharmacies and the Canadian Institute for Health Information’s (CIHI) National System for Incident Reporting.
Medications Frequently Reported (ISMP, 2020)
- Community settings: methadone, metformin, levothyroxine
- Hospital settings: Insulin, hydromorphone, morphine
- Home and community care: hydromorphone, methotrexate, bisoprolol
- Long-term care: hydromorphone, fentanyl, insulin
For more information, go to the ISMP Canada site: Medications most frequently reported in harm incidents over the past 5 years (2015-2020). https://ismpcanada.ca/wp-content/uploads/Figure-4-ISMPCSB2020-i11-Medications-Reported-Harm-.jpg
Medications Most Frequently Reported in Harm Incidents over the Past 5 Years (2015-2020)
This data shows that medication incidents are not just occurring in acute care settings but in all client care settings and with similar frequency. Hydromorphone was among the top three drugs in three out of four settings reporting severe harm or death reports.
Other drugs that had a higher incidence of severe harm or death incidents included hydromorphone, morphine, methotrexate, methadone, and lorazepam; all of which are considered high-alert medications.
The ISMP (2024) has published a list of High-Alert Medication in Acute Care Settings. These medications are a heightened risk due to the risk of harm if used in error, and as such, require special safeguards to minimize the risk. For example, safeguards can include limiting access to high-alert medications or using clinical decision support tools. High-risk medication can be remembered using the mnemonic “A PINCH.” Table 3.7.1 describe the medications included in the “A PINCH” mnemonic.
| High-Risk Medication Group | Medication Examples |
|---|---|
| A: Anti-infective |
|
| P: Potassium and other electrolytes |
|
| I: Insulin |
|
| N: Narcotics & Other Sedatives |
|
| C: Chemotherapeutic Agents |
|
| H: Heparin & Antithrombolytics |
|
Note: For the complete list, go to ISMP’s (2024) List of High-Alert Medications in Acute Care Settings [PDF].
Error-Prone Abbreviations
Error-prone abbreviations are abbreviations, symbols, and dose designations that have been reported through the ISMP National Medication Errors Reporting Program as being frequently misinterpreted and involved in harmful medication errors. ISMP Canada’s (2017) Do Not Use: Dangerous Abbreviations, Symbols, and Dose Designations should never be used when communicating medical information. Some examples of abbreviations that were commonly used that should now be avoided are qd, qod, qhs, BID, QID, D/C, subq, and APAP.
Strategies to avoid mistakes related to error-prone abbreviations include not using these abbreviations in medical documentation. Furthermore, if a nurse receives a prescription containing an error-prone abbreviation, it should be clarified with the provider and the order rewritten without the abbreviation.
Do Not Crush List
The ISMP (2022) also maintains a list of oral dosage medication that should not be crushed, commonly referred to as the do not crush list. These medications are typically extended-release formulations.
Strategies for preventing harm related to oral medication that should not be crushed include requesting an order for a liquid form or a different route if the client cannot safely swallow the pill form.
Look-Alike and Sound-Alike (LASA) Drugs
ISMP Canada maintains a list of drug names containing look-alike and sound-alike name pairs.
For example:
- captopril and carvedilol, and
- dimenhyDRINATE and diphenhyDRAMINE.

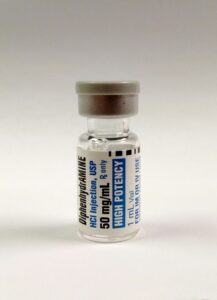
Figure 3.7b DimenhyDRINATE and diphenhyDRAMINE are two meds with similar sounding names, with high risk for a med error.
(Onnifrey/Wikimedia Commons) CC-BY-SA-4.0) (dennissylvesterhurd/flickr) CC BY-SA 1.0
These medications require special safeguards to reduce the risk of errors and minimize harm. For a full list of these medications, you can review the following resource ISMP’s (2019) Look Alike-Sound Alike List of Medications.
Safeguards may include:
- using both the brand and generic names on prescriptions and labels;
- including the purpose of the medication on prescriptions;
- changing the appearance of look-alike product names to draw attention to their dissimilarities; or
- configuring computer selection screens to prevent look-alike names from appearing consecutively. (ISMP Canada, 2019)
Strategies for safe administration of high-alert medication include:
- standardizing the ordering, storage, preparation, and administration of these products;
- improving access to information about these drugs;
- employing clinical decision support and automated alerts; or
- using redundancies such as automated or independent double checks when necessary. (ISMP Canada, 2019)
Lastly, the above information from ISMP may differ from your agency. Always check with your area of practice for unit specific guidelines or requirements to avoid medication errors.
References
Institute for Safe Medication Practices. (2019). List of confused drug names. https://web.archive.org/web/20220320141944/https://www.ismp.org/recommendations/confused-drug-names-list
Institute for Safe Medication Practices. (2022). Oral dosage forms that should not be crushed. Internet Archive. https://web.archive.org/web/20220301174924/https://www.ismp.org/recommendations/do-not-crush
Institute for Safe Medication Practices. (2024). ISMP list of high-alert medications in acute care settings [PDF]. https://www.ismp.org/system/files/resources/2024-01/ISMP_HighAlert_AcuteCare_List_010924_MS5760.pdf
Institute for Safe Medication Practices Canada. (2018). Do not use: Dangerous abbreviation, symbols and dose designations [PDF]. https://www.ismp-canada.org/download/ISMPCanadaListOfDangerousAbbreviations.pdf
Institute for Safe Medication Practices Canada. (2020). Medications most frequently reported in harm incidents over the past 5 years (2015–2020). https://ismpcanada.ca/bulletin/medications-most-frequently-reported-in-harm-incidents-over-the-past-5-years-2015-2020-2/
Media Attributions
- Figure 3.7a. Institute for Safe Medication Practices Canada.
- Figure 3.7b DimenhyDRINATE and diphenhyDRAMINE are two meds with similar sounding names. med.(Onnifrey/Wikimedia Commons) CC-BY-SA-4.0) https://commons.wikimedia.org/wiki/File:Diphenhydramine_1_(cropped).jpg (dennis sylvesterhurd/flickr) CC BY-SA 1.0 https://flickr.com/photos/43296902@N00/26879845435

