1.7 Pharmacodynamics
Complex Interactions
So far, we have learned the importance of pharmacokinetics in describing how the body absorbs, moves, processes, and eliminates a medication. Now, let us consider a drug’s impact on the body, which is a series of complex interactions known as pharmacodynamics. Pharmacodynamics is simply what the drug does to the body. It is a crucial part of pharmacology as it looks at the safety of the drug and identifies dosage ranges to achieve a therapeutic effect.
How a drug responds in a body is not always predictable, with some clients eliciting the desired response while others may have an adverse effect or no effect at all. The study of pharmacodynamics helps practitioners understand how effective a drug might be, also known as its efficacy. It tells us how strong a drug response might be, also known as its potency. It also helps our understanding of the relationship between pharmacokinetics and pharmacodynamics and see how both influence dosing, effectiveness, and adverse effects.
Pharmacodynamics is crucial to understand the mechanisms of action, dose-response relationships, and the variability in individual drug responses. Pharmacodynamics explores how drugs exert their effects on the body, specifically the specific molecular targets referred to as receptors, enzymes, and ion channels. These interactions can either stimulate or inhibit the function of these targets, resulting in physiological changes (Hook, 2023).
Drug Receptor Interactions — Key Points
- In general, drugs or substances (chemicals) will not act unless they induce a reaction with a living organism. Drugs also cannot make a cell do something it was not designed to do. The effects of the drug will depend on the tissues or cells targeted by the drug.
- Drugs induce a reaction in several ways: either they modify the rate that the cell functions or modify the strength of function of that cell or tissue. Therefore, they only produce an effect by interacting with other chemicals or receptors.
- Drugs can bind or interact with a variety of chemicals or proteins: these are receptors, transporters, ion channels, enzymes, nucleic acids or lipids.
- Receptors families: Receptors are proteins, but they vary in their structure. They can be cell membrane-embedded enzymes, ligand-gated ion channels, G-protein-coupled receptor systems, or transcription factors.
- Drug-receptor binding can be reversible or irreversible. Most either mimic or inhibit normal physiological processes or pathological processes.
- Efficacy and potency of a drug-receptor interaction are key terms for pharmacodynamics.
Drug Interactions
The effects of a drug depend on the cells or tissue targeted by the drug. Once the drug is at the site of action, it can only mimic or block the actions of the endogenous molecules. A drug cannot make a cell do something it was not designed to do. It can only increase or decrease the physiological activity normally controlled by that receptor. Three ways which drugs do this are receptor, enzyme, and non-selective interactions.
Receptor Interaction
Receptor interaction involves the selective joining of the drug molecule with a reactive site on the surface of a cell or tissue. This is usually a protein structure within the cell membrane. As previously mentioned, there are four primary receptor families (G-coupled, ligand-gated ion channels, cell membrane embedded, and transcription factors). Although each are distinct in their function and processes with drug binding, all work similarly with a drug (a chemical) interacting with a receptor (chemical site) to produce a physiological effect.
Once the substance (drug or chemical) binds to and interacts with the receptor, a pharmacological response is produced.
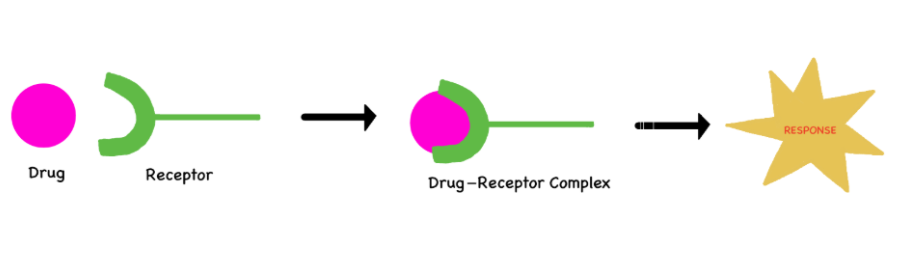
The binding of a drug to a receptor is usually reversible. This drug-receptor binding can only mimic or block the action of the endogenous regulatory molecule. The drug with the best fit or greatest affinity to that receptor will elicit a greater response from the cell.
Some drugs interact with a receptor and activate it fully. These are called agonists:
- They can only mimic or enhance what the cell was intended to do.
- They will produce the same response as the endogenous chemical.
- Examples: Many neurotransmitters (histamine, norepinephrine), hormones, and drugs (opiates, benzodiazepines) are agonists.
Some drug molecules only partially activate the receptor (partial agonist):
- The response will be lesser than or weaker than an agonist.
- Example: Buprenorphine is sometimes prescribed for opioid addiction. The degree of pain relief (and other euphoric effects) will be much lower than a full agonist, such as morphine.
Other drugs block the receptor to produce no action at all (antagonist):
- They block receptor activity with the effect most evident in the presence of an agonist. They will occupy the receptor and prevent the endogenous chemical from acting.
- The response of the antagonist is dependent on how much agonist drug is present.
- Example: Naloxone is given for respiratory depression from opioid overdose. Naloxone, which has a high affinity to the mu receptors, will occupy free mu receptors as well as replace the opioid from the mu receptor it is occupying. If Naloxone was given when no opioid drug was given previously, then naloxone would produce no response.
(Marino, Jamal, & Zito, 2023; Rosow, Standaert, & Strichartz, n.d.).
Figure 1.7b represents the drug-receptor complex reactions. Before the drug is given, the body’s own endogenous molecule binds with a receptor site on a cell and elicits a response. The second diagram, the agonist drug, with a similar composition as the endogenous molecule, is able to produce the same response. The third diagram, the antagonist drug binds to the receptor and only blocks the agonist’s actions. No response will occur.

Figure 1.7b Agonist and antagonist drug-receptor responses (Dolleyj/Wikimedia Commons) CC BY-SA 3.0
In the following graph, the full agonist produces a greater response or greater activity. The partial agonist, with a similar concentration, is not able to produce the same activity.
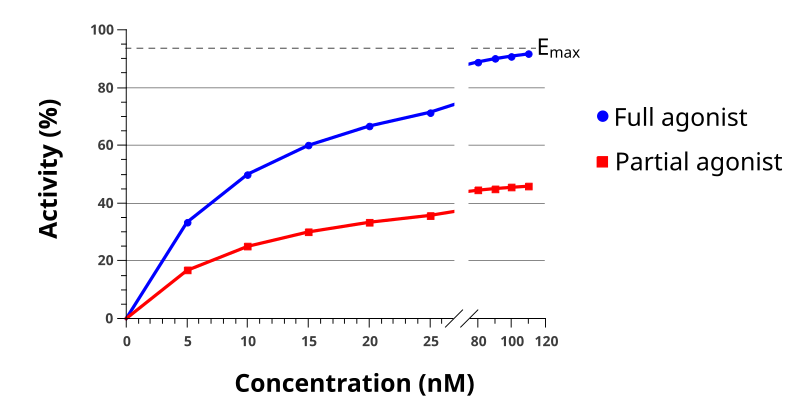
File: Agonist full and partial.svg. . https://commons.wikimedia.org/wiki/File:Agonist_full_and_partial.svg Creative CommonsAttribution-Share Alike 3.0 Unported license
 Critical Thinking in Action
Critical Thinking in Action
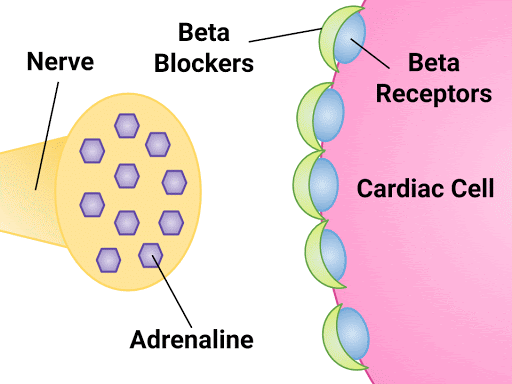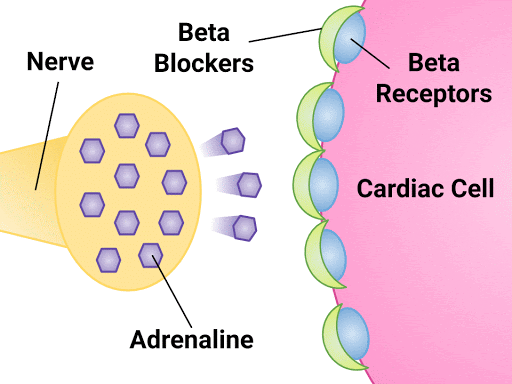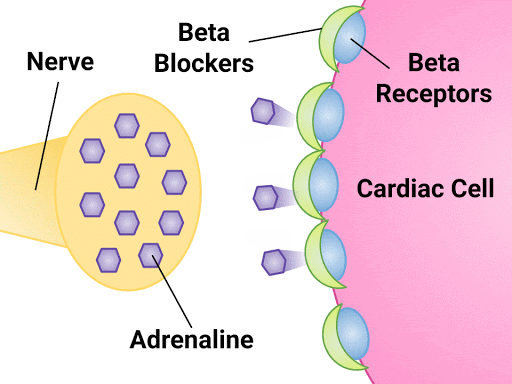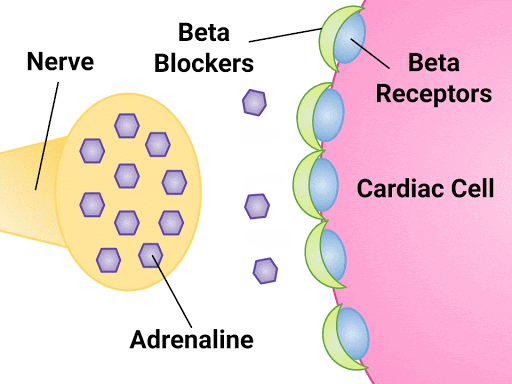
A client has heart failure and needs a medication to lower their heart rate.
Recall that beta-1 receptors are located on smooth muscle cells in blood vessels and on heart muscle cells. The hormone adrenaline is released from the nerve cell, binding to the receptors on the blood vessels or heart and tightening up the blood vessel (leading to increased BP) or elevating the heart rate.
The prescriber orders the cardiac medication atenolol, a beta-1 receptor antagonist.
Atenolol will bind to the beta-1 receptors on the heart leading to:
- Slower contractions of the heart (negative inotropic effect), thus causing the heart muscle to work less;
- Heart rate decreases (negative chronotropic effect); and
- Slower heart conduction (negative dromotropic effect).
The nurse will anticipate the effects of atenolol and be able to assess and monitor appropriately.
Note: Answers to the Clinical Reasoning and Critical Thinking activities can be found in the “Answer Key” sections at the end of the book.
Agonistic and antagonistic effects on receptors are further discussed in the Autonomic Nervous System chapter.
Enzyme Interaction
Enzymes are substances that catalyze nearly every biochemical reaction in a cell. The drug chemically binds to an enzyme molecule, thereby changing the physiological response.
The drug can enhance, inhibit, or deactivate the enzyme, but inhibit is the most common response. These interactions can be reversible or irreversible.
Example: Cyclooxygenase (COX) inhibitors, such as non-steroidal anti-inflammatory drugs (NSAIDs), act on one or both of the isozymes COX-1 and COX-2. The inhibition of these pathways leads to pain relief and a decrease in inflammation.
Example: Drugs can also be activated by the enzyme reaction. Metronidazole (anti-infective) enters a bacteria cell, where it is activated by bacterial enzymes, and then kills the bacteria.
Non-specific Interactions
Some drugs physically interfere with or chemically alter cell processes and cell membranes. They do not target or interact with receptors or enzymes. This results in an improperly formed cell wall, which leads to cell lysis or, if it lacks the needed energy, cell starvation.
Example: Antibiotics that affect the bacterial cell walls. Beta-lactams such as penicillin interfere with the structure of peptidoglycan (component of the bacterial cell wall), leading to cell wall lysis.
Example: Mannitol, an osmotic diuretic, increases the blood plasma osmolarity leading to cells (brain primarily) becoming dehydrated as water moves to the bloodstream.
Example: Antacids modify stomach acid (pH) through a chemical reaction:
[latex]\mathrm{Mg}(\mathrm{OH})_2+2\mathrm{HCl}\rightarrow2\mathrm{H}_2\mathrm{O}+\mathrm{MgCl}_2[/latex]
This chemical reaction neutralizes stomach acid and relives symptoms.
Important Terms — Affinity, Potency, Efficacy, and Selectivity
If a drug has a strong affinity to a receptor, does that mean it will be the most effective? Are all drugs equally selective? What happens when two drugs with similar actions are given together; will it always end up as a bigger response? The answer to these questions helps nurses understand why one drug is chosen over another drug and what we need to be monitoring to ensure client safety.
Affinity is how attracted the drug is to the receptor. A higher affinity of a drug molecule to a receptor means a tighter bond or attraction. If you have two drugs that can bind to a receptor, the drug with the higher affinity will occupy more of the receptor sites.
Potency is how much drug is needed to produce an effect. It is often measured as the concentration of the drug needed to achieve 50% of the drug’s maximal effect (EC50 effective concentration 50). A drug with a high affinity to a receptor are more potent as they can bind to receptors at low concentrations.
Efficacy (maximum effect E max) refers to a drug’s maximal effect. It is the drug’s capacity to produce an effect or activate a receptor, regardless of the dose. It measures how well a drug works. Note that just because a drug occupies a receptor, does not mean it will produce a maximum response. It can produce a complete response, no response or a partial response.
Intrinsic activity, related to efficacy, is the drug’s ability to activate a receptor and produce a response in comparison to a full agonist.
Selectivity is how likely it is for a drug to only affect its target site. A highly selective drug will only create a response in a given target site—it causes a change in certain cells without affecting other cells. It is desirable to have a drug that is highly selective so only the desired effect is achieved. Drugs that bind with receptors beyond the target site will create unwanted effects or side effects. For example, morphine binds to mu and kappa pain receptors in the CNS to alleviate pain, but it also binds to other pain receptors (kappa, delta) outside the CNS and other non-selective receptor effects in the CNS and peripheries. This leads to side effects such as nausea or respiratory depression.
Efficacy of Two Drugs
In the diagram below, we have two drugs. Drug A is shown to have a bigger effect or response than with Drug B. Drug A is more efficacious. Drugs with a higher efficacy are often more desirable as they produce a higher therapeutic response (Yartsev, 2015).
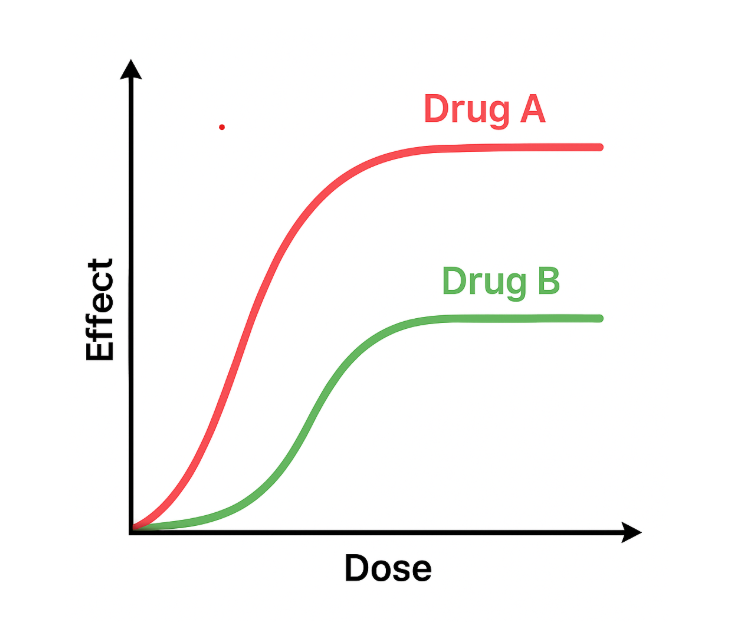
Figure 1.7e Comparison of efficacy between two drugs (Sheila Odubote/ TRU Open Press)
Example in Practice:
A client has congestive heart failure and needs a diuretic to treat fluid overload. Let us compare two diuretics:
- Furosemide, Drug A, is a loop diuretic that eliminates sodium and water through the ascending loop of Henle in the kidneys, leading to an fluid loss, less intravascular volume and a lower BP.
- Hydrochlorothiazide, Drug B, is a thiazide diuretic that also causes water loss through the kidneys, but its actions are less powerful and effective compared to Furosemide.
Thus, Furosemide has greater efficacy than hydrochlorothiazide.
In this diagram, figure 1.7f, Both Drug A and Drug B achieve the same maximum effect, i.e., they have equal efficacy. However, Drug A achieves this effect at a lower dose, so Drug A has a higher potency than Drug B.
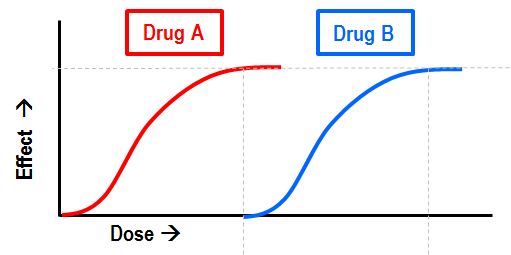
Example In Practice:
A client needs pain control and two opioid drug options are compared. Hydromorphone 1.5 mg po is equivalent to Morphine 10 mg po. Hydromorphone is more potent than morphine although both are equally effective.
Mechanism of Action
The interaction between a drug and a receptor is the mechanism of action (MOA), or “how the drug works.” This is the way that a drug produces its pharmacological effect. Sometimes, drugs affect target cells through enzymes, by changing cell function, or by changing the cellular structure itself (Barbour-Taylor et al., 2024).
Example in Practice:
Let us compare two pain medications:
- Morphine, an opioid, acts directly upon mu and kappa receptors in the central nervous system and alters the perception of pain.
- Ibuprofen, an anti-inflammatory and pain reliever, works on the cyclo-oxygenase pathway by blocking prostaglandin synthesis.
Although both drugs are pain relievers, their actions affect different receptors on different pathways or cells. Why is this important? Nurses need to understand the MOA of the drugs they administer to be able to anticipate the desired effect (strong, moderate, or mild pain relief), side effects (nausea, gastric irritation, or pruritis), and adverse effects (respiratory depression or gastric bleed).
The mechanism of action is known for most medications, which helps with understanding the desired effects as well as side effects and adverse reactions. But for some medications, it is either not clearly understood or unknown. For example, the MOA for acetaminophen, a common pain reliever, is not completely understood, and neither is the weight gain associated with antipsychotic medications.
The principles discussed here are not a comprehensive list but an introduction to the ways some drugs interact with cells. Nurses need to have a firm grasp of these basic principles and apply them for safe medication administration.
Putting It All Together
A client has 8/10 pain and is given Morphine IV. Morphine is a full agonist that will bind to the mu receptors in the CNS to relieve pain. This client’s pain is relieved, but their breathing becomes shallow and depressed leading to respiratory depression.
The nurse gives naloxone, which is an opioid antagonist. Naloxone has a very strong affinity to mu receptors, and in the presence of the agonist morphine, will bind to the mu receptors, displacing morphine. By giving naloxone, the pain receptors are now not occupied by an agonist and the pain returns. But respirations will improve, which is the intended effect with giving naloxone.
While a full agonist produces the maximal response at that receptor when they bind, partial agonists bind to the same receptor but produce a smaller response, even when all receptors are occupied. An example is buprenorphine, which is used to treat opioid addiction. It can relieve pain like a full agonist but it has a ceiling effect so the risk of respiratory depression is minimal.
In summary, nurses need to have an understanding of the mechanism of action of medications they are giving to ensure they can effectively assess, administer, and monitor for therapeutic and side effects.
Learning Activities
Clinical Reasoning and Decision-Making 1.7
1. A nurse is reading in the drug manual that a drug is potent. The nurse considers what potent means, and thinks it refers to the drug highly likely to cause adverse or toxic effects. Is this nurse correct? Explain.
Note: Answers to the Clinical Reasoning and Critical Thinking activities can be found in the “Answer Key” sections at the end of the book.
References
Barbour-Taylor, T., Mueller (Sabato), L., Paris, D., & Weaver, D. (2024). 2.2 Pharmacokinetics and pharmacodynamics. In Pharmacology for nurses. OpenStax. https://openstax.org/books/pharmacology/pages/2-2-pharmacokinetics-and-pharmacodynamics. Access for free at https://openstax.org/books/pharmacology/pages/1-introduction
Farinde, A. (2023). Overview of pharmacodynamics. Merck Manual. https://www.msdmanuals.com/professional/clinical-pharmacology/pharmacodynamics/overview-of-pharmacodynamics
Hook M. (2023). Significance and its characteristics of pharmacodynamics. Journal of Basic Clinical Pharmacology, 14(5), 313–314. https://www.jbclinpharm.org/articles/significance-and-its-characteristics-of-pharmacodynamics-12567.html
Marino, M., Jamal, Z., & Zito, P. M. (2023). Pharmacodynamics. StatPearls. Retrieved October 20, 2025, from https://www.ncbi.nlm.nih.gov/books/NBK507791/
Rosow, C., Standaert, D. & Strichartz, G. (n.d.). Agonist and Antagonist Actions. Principles of Pharmacology LibreTexts. CC BY-NC-SA 4.0
Thomas, A., & Young, S. (2008). An introduction to pharmacodynamics. Practice Nursing, 19(12), 596–600. https://doi.org/10.12968/pnur.2008.19.12.31852
Media Attributions
- Figure 1.7a Drug-Receptor complex. (Sheila Odubote/ TRU Open Press). CC BY-NC-SA 4.0
- Figure 1.7b Agonist & Antagonist.jpg File:Agonist & Antagonist.jpg – Wikimedia Commons by Dolleyj, Wikimedia Commons, CC BY-SA 3.0
- Figure 1.7c Agonist full and partial.svg. . https://commons.wikimedia.org/wiki/File:Agonist_full_and_partial.svg
- Figure 1.7d Mechanism of Action” by Dominic Slausen at Chippewa Valley Technical College is licensed under CC BY 4.0 license.
- Figure 1.7e Comparison of efficacy between two drugs (Sheila Odubote/ TRU Open Press)
- Figure 1.7f Efficacy and potency between two drugs (Sheila Odubote/ TRU Open Press).
The study of how drugs act at target sites of action in the body.

