1.4 Pharmacokinetics — Distribution
The second stage of pharmacokinetics is the process known as drug distribution. Distribution is the process by which medication is dispersed throughout the body via the bloodstream. Once a drug enters into systemic circulation by absorption or direct administration, it must be distributed into interstitial and intracellular fluids to get to the target cells.
Factors Impacting Drug Distribution
The distribution of a drug throughout the body is dependent on a number of client-related factors. These include:
- blood flow to the tissues,
- plasma protein binding,
- body composition (fat and water),
- health issues affecting albumin,
- blood-brain barrier, and
- if the client is pregnant, the placental barrier.
All of these factors are inter-connected although we will examine each separately. All of these factors will have an impact on the volume of distribution, which is a reference point for plasma drug concentration.
Blood Flow to the Tissues
During the first phase of distribution, drugs are carried by the blood to organs and tissues in the body. Blood flow is one of the primary determinants of drug distribution. In a healthy person, tissues are well perfused with good blood flow, so drug distribution is not impacted. Blood flow is the primary indicator of the rate and amount of drug uptake at target sites. In certain health issues. low perfusion can impact the ability of a drug to reach its target site. For example, a client in septic shock may have reduced perfusion to their extremities leading to mottling and less blood flow to some organs to maintain hemodynamic stability. Certain conditions such as tumours or abscesses can also affect drug therapy. An abscess has no internal blood supply, so antibiotic therapy cannot reach the pathogens easily. For solid tumours, some are resistant to drug therapy due to the limited blood supply (Burcham & Rosenthal, 2019).
Drugs carried by the bloodstream will penetrate different organs or tissues at different rates. Organs with high perfusion, such as the heart, liver and kidneys will have higher distribution of drugs than bones, adipose tissue and the skin, which have lower distribution. For the latter organs, this characteristic is only a potential challenge if high concentrations of drugs are needed to be delivered to these areas. For example, a client with osteomyelitis may be on a longer course of antibiotics due to the difficulty penetrating into the bone.
Membrane permeability or membrane barriers also have an important role in drug distribution. Some membranes are more permeable to drugs so the drug can move easily from the bloodstream into the tissues. See below for the discussion on lipophilic and hydrophilic drugs.
Body Composition — Water and Fat Composition
The body’s water composition reduces as we age. Young children will have a higher percentage of body water compared to older adults; however, intracellular water is stable.
Individuals also have varying degrees of fat composition, either due to age, gender, genetics, or lifestyle. For clients with a higher fat composition, lipophilic drugs will have a larger volume of distribution to fat tissues, resulting in a longer duration of action and lower blood concentrations. Conversely, hydrophilic drugs have a small volume of distribution and higher blood concentrations.
Health Issues Impacting Albumin Levels
Albumin levels can be impacted by various disease or health states, such as hypovolemia, malnutrition, inflammation, burns, or third spacing due to sepsis. In these cases of low serum albumin levels or hypoalbuminemia, the drug molecule has less sites to bind to, so more of the drug is circulating in the bloodstream and reaching target sites.
For clients who have or are at risk of hypoalbuminemia, nurses can anticipate that serum albumin levels will be drawn before drug therapy and at intervals throughout the client’s treatment. Nurses can also anticipate lower drug dosages until a client’s nutritional status is corrected if due to malnutrition, or replacing albumin through IV administration to correct the deficiency. This is also sometimes done for clients with liver disease.
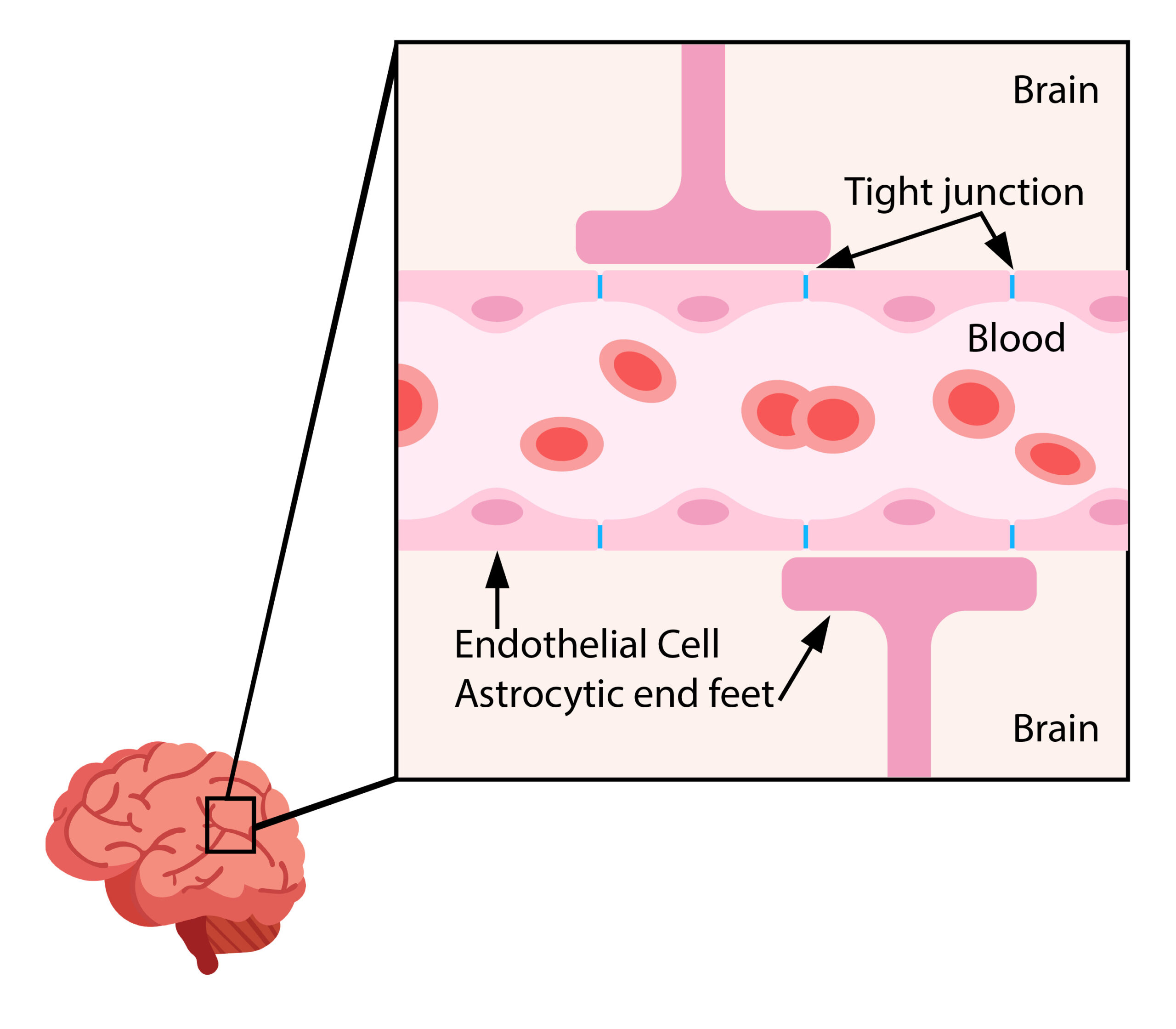
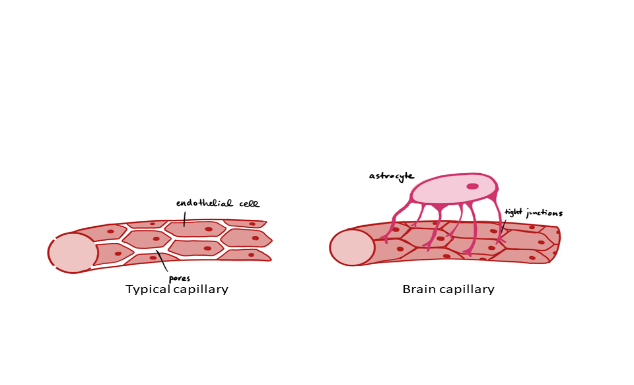
Blood-Brain Barrier
Medications destined for the central nervous system (the brain and spinal cord) face an even larger hurdle than protein binding; they must also pass through a nearly impenetrable barricade called the blood-brain barrier (BBB).
There are 3 main factors that prevent drugs or other substances from crossing the BBB:
- Endothelial cells form a physical barrier with tight junctions between cells, reducing passive diffusion.
- An enzymatic barrier breaks down certain molecules that may be in transit across the cell membrane.
- Astrocytes are spread out along the cell membrane, and their end-feet extend down into the endothelial cells, forming a physical barrier.
Figure 1.4a Blood brain barrier. (TRU Open press). This diagram shows the capillary with tight junctions between endothelial cells and the astrocyte feet that prevent molecules from entering the brain.
To understand more about the Blood Brain Barrier: 2 minutes Neuroscience: Blood Brain Barrier. YouTube: Neuroscientifically Challenged https://www.youtube.com/watch?v=e9sN9gOEdG4
Making it through the blood brain barrier:
How can nutrients get into the brain?
- Glucose and amino acids (small polar solutes) need transport proteins or carriers to cross the BBB. Small lipophilic molecules and gases, such as oxygen and carbon dioxide, can diffuse across the lipid membrane.
How can drugs get into the brain?
- Only certain medications that are made of lipids (fats) or have a “carrier” can get through the BBB. Research scientists have devised ways for certain medications to penetrate the BBB. An example of this is the brand-named medication Sinemet®, which is a combination of two drugs: carbidopa and levodopa. Carbidopa is designed to carry the levodopa medication across the BBB, where it enters the brain and is converted into dopamine to exert its effect on Parkinson’s disease symptoms.
- Some medications inadvertently bypass the BBB and impact an individual’s central nervous system function. For example, diphenhydramine (Benadryl®) is an antihistamine used to decrease allergy symptoms; however, it can also cross the BBB, depress the central nervous system, and cause the side effect of drowsiness. In the case of a person who has difficulty falling asleep, this drowsy side effect may be useful, but for another person it may be problematic as they try to safely carry out daily activities.
Placental Barrier
It is always important to consider the effects of medication for clients who are pregnant or may become pregnant. The placenta is permeable to some medications, while others have not been specifically studied in pregnant clients. Most drugs can cross the placental barrier. Lipid-soluble nonionized drugs cross the placenta, and these compounds can pass from maternal into fetal circulation. Only drugs that are protein-bound, ionized, or highly polar cannot move easily across the placental barrier. As the pregnancy progresses, physiological changes occur that can change the distribution of drugs. As the plasma volume expands, this leads to an overall diluting effect on plasma proteins. Adipose tissue increases, which can increase the volume of distribution of medications, leading to a more prolonged effect.
During any trimester, some drugs can cause harm to the unborn fetus. In the first trimester, some drugs can cause birth defects (congenital anomalies) that can range from physical to cognitive anomalies, such as cleft palate, spina bifida, metabolic disorders, or heart defects. Other drugs can have an effect later in the pregnancy, resulting in low birth weight and premature labour.
Therefore, it is imperative to always consult a healthcare provider regarding the safety of medications during pregnancy. Further, it is also important to ensure clients are aware that any medication, either prescribed, OTC, or a natural health remedy, is first reviewed by their practitioner. This imperative is assumed in the remaining chapters discussing medication classes, and nurses should always check the most recent, evidence-based drug references before administering medications during pregnancy.
Drug-Related Factors
Distribution is also impacted by drug-related factors such as the molecular structure of the drug, lipid solubility, and plasma protein and tissue binding.
Drugs leave the vascular system unimpeded by passing through the pores in the capillary wall. As mentioned above, drug distribution to various organs or tissues is determined by blood flow to that area, but it is also dependent on the molecular structure of the drug.
Lipid-Soluble vs. Water-Soluble Drugs
A drug’s permeability across cell membranes is a key factor that influences the absorption and distribution. This is because if a drug wants to reach to the systemic circulation, it needs to cross several semipermeable cell membranes first.
Lipid-soluble drugs are distributed more completely to body tissues due to their ability to be quickly absorbed, mix with the blood, cross membranes, and then localize to tissues (Adams et al, 2018). Therefore, lipid-soluble drugs will have a higher volume of distribution and a lower blood concentration. In comparison, highly water-soluble drugs will have a smaller volume of distribution and high blood concentration. Refer to the previous section, 1.1 Pharmacological Principles, for a review on lipid and water soluble drugs.
Plasma Protein and Tissue Binding
In the bloodstream, drugs are transported partly as a free (unbound) drug and partly as being bound reversibly to plasma proteins (drug-protein complex). Albumin is the most abundant protein in the blood and the protein that most drugs bind to, although other proteins include lipoproteins and alpha-1 glycoprotein. Only unbound drugs can passively diffuse to the extravascular sites where the target cells are located. Unbound drug concentrations in the vascular system typically determine the drug concentration at the target site. This determines the efficacy of the drug.
The large drug-protein complexes circulate in the bloodstream until they are displaced or become unbound, which leaves the drug molecule small enough to pass through the capillary beds and reach the target sites. If this did not occur, the bound molecules could not reach the target sites, leading to a prolonged distribution phase and increased drug half-life.
Albumin
Albumin is a large protein molecule in the plasma, so large that it primarily remains intravascularly. It has no transport system and is too large to squeeze through the pores of the capillary wall.
Normal Albumin level: 34–50 g/L
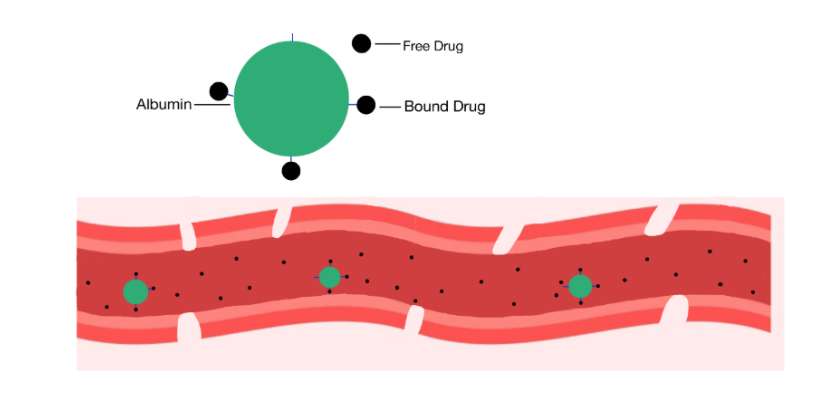
The major drug-binding protein in serum is albumin. Drugs exist in peripheral circulation as free (unbound) and bound to protein forms. Only free drugs are capable of crossing plasma membranes and binding with receptors for their intended action.
Displacement of Drugs From Albumin
The plasma protein albumin only has so many site for drug molecules to bind to, so they compete with each other for those sites. Drug molecules with a higher affinity for albumin will competitively bind to plasma proteins first or will displace other drugs from the albumin binding sites. This leads to an increased concentration of the secondary drug in the bloodstream and more of the drug reaching the target site. This can lead to increased therapeutic or toxic drug levels. This is called drug-drug interaction. Drug-drug interaction occurs when a drug decreases or increases the response of another drug that is concurrently administered.

Think of protein-binding like a bus stop (see Figure 1.4d). Many passengers (or medication molecules) want to take a ride on the bus. Everyone is eager to get to their destination and interested in finding a seat. Some passengers are stronger and will get in the seats first (like drug molecules with greater protein-binding ability bind to the protein). Sometimes, there may not be enough seats on the bus, and some passengers are left at the bus stop. The passengers (medication molecules) left behind are “free” to move around and walk to their destination. They may strike out on their own and get “snatched” (connected to a target receptor site) while on foot.
In a similar way, “free” drug particles that are not protein-bound are circulating in the bloodstream and connecting in a predictable fashion to receptor sites that have an affinity for that particular drug. These active drug molecules that did not bind to the protein (like those passengers that were unable to get a seat on the bus) will produce the first effect in the body. Over time, the medication molecules bound to the protein (like the passengers with seats on the bus) will get off the bus, start walking around, and get “snatched” by the receptor site that has affinity for them.
What Makes a Drug Molecule Bind to the Albumin Receptor?
A drug molecule binds to an albumin receptor because of affinity. A strong affinity (attraction) will have a large number of drug molecules bound to albumin, leaving a small percentage free to go to target sites. For example, the anticoagulant warfarin has a high affinity for albumin, up to 99% of the molecules are bound. The antibiotic Gentamycin has a weak affinity for albumin, so most of the drug molecule is unbound, resulting in more of the drug reaching the target site.
Are all drugs bound to albumin? The protein binding of a drug can be low, moderate, or high, whereas for some drugs such as lithium, the drug is not bound to serum protein at all.
Example in Practice:
Peter has atrial fibrillation. To manage the risk of blood clots, he takes the anticoagulant warfarin. Peter has been following the treatment plan, but he recently developed heartburn. He bought OTC cimetidine (an anti-ulcer medication H2 receptor antagonist) and started taking it over the last week. Is it alright for him to take both of these meds? It is safe?
Cimetidine has a very strong affinity for albumin, so it has a high likelihood to displace highly protein-bound drugs, increasing free drug levels and the risk of toxicity. In this case, warfarin is a highly protein bound drug, and cimetidine can displace warfarin. This will lead to more warfarin molecules free in the bloodstream and increase the risk of hemorrhage. This poses a safety concern for Peter. He should not be taking any other medications without the advice of his prescriber and without close monitoring for bleeding.
Theoretical Volume of Distribution (Vd)
The volume of distribution (Vd) is used to describe the potential volume within various areas where drugs may be distributed. These fluid compartments can be the plasma, extracellular water, total body water, or other organs and tissues. Vd has nothing to do with the actual volume of fluid in the body; instead, it relates the distribution of the drug in the body. Each drug has its own unique distribution in the body: some are distributed mostly into fat tissue, some remain in extracellular fluid, and some are bound to tissues. Vd provides a reference point for the plasma concentration of a drug.
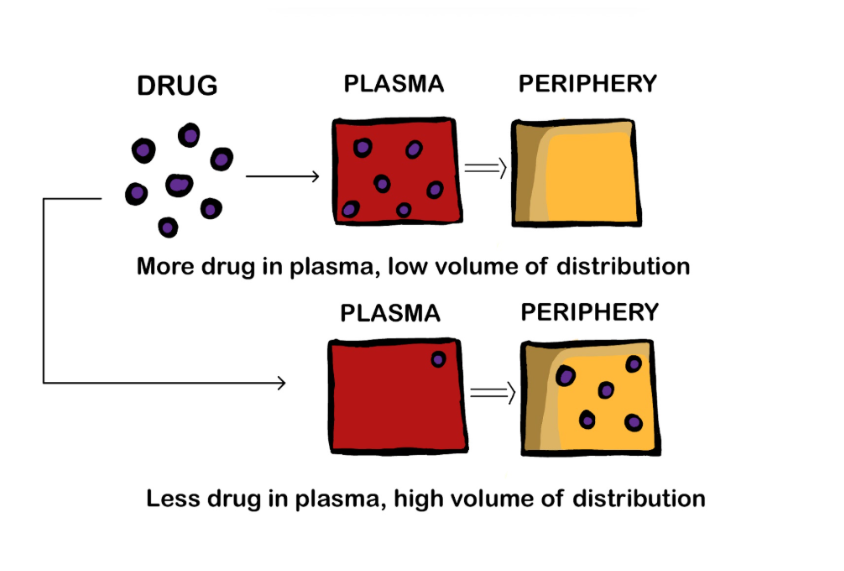
Figure 1.4e Volume of distribution. (Sheila Odubote/ TRU Open Press). If most of the drug stays in the plasma and very little goes to the periphery (fat stores, interstitial fluids) then the drug has a low volume of distribution. If most of the drug moves to the periphery such as adipose tissue, then the drug has a high Vd and might be lipophilic.
Drugs do not distribute equally. Each organ or body tissue will receive different amounts of a drug. Many factors can affect distribution including blood flow to the tissues, protein binding, and lipid solubility. Client factors include age, disease states, drug interactions, obesity, and malnutrition.
For example, for a highly tissue-bound drug, very little of the drug will remain in the bloodstream, and therefore the plasma concentration is low and Vd is high. Highly protein-bound drugs, such as warfarin or acetylsalicylic acid, will have a small volume of distribution.
So what does this mean in practice? Drugs with a high Vd may take longer to be eliminated from the body. They have a longer t½ and therefore their effects, including therapeutic and side effects, last for a longer period of time.
The Plasma Half-Life of a Drug
Half-life (t½) is the amount of time it takes for half of the medication to be eliminated from the body. Half-life directly correlates to the duration of the therapeutic effect of a medication. At four half-lives, only 6.25% of a drug is left in the body, which is at sub-therapeutic levels. At five half-lives, 97% of a drug will be eliminated.
Many medications are classified in terms of their half-life as it helps determine dose scheduling and when a steady state will occur. A steady state is when the amount of the drug removed by elimination is equal to the amount of the drug absorbed with each dose. If a drug has a long half-life, it will take longer to reach a steady state. See the unit Examining the Drugs Effects for further discussion on this topic.
Distribution of a medication can also cause unintended adverse effects or side effects. Drugs are designed to primarily cause one effect, meaning they bind more strongly to one specific receptor site and predictably cause or block an action. However, side effects can occur when the drug binds to other sites in addition to the target tissue, causing secondary side effects. These side effects can range from tolerable to unacceptable, resulting in the discontinuation of the medication. For example, a person might take the pain reliever ibuprofen (Advil) to treat a sore leg muscle; although the pain may be subsequently relieved, the side effect of stomach irritation may cause the person to stop taking it.
 Critical Thinking Out Loud
Critical Thinking Out Loud
Manjot is at 36-week gestation and has had a healthy pregnancy. In the event that Manjot needs to be given medications, which effect, if any, will her late-term pregnancy have on drug distribution?
Points to consider:
- Late-term pregnancy
- Concern with drug effects on fetus (placenta drug transfer)
- Concern if early delivery and effect on newborn
- Consider/anticipate dosage changes of medications related to pregnancy
The last term of pregnancy can impact albumin levels. The plasma volume expands, which can lead to hypoalbuminemia. Adipose tissue also increases, causing a larger distribution of the medication and leading to a prolonged duration of action and longer t ½ for medications.
Solution:
- Test serum albumin levels to determine if Manjot has hypoalbuminemia.
- Anticipate lower drug dosages. To avoid toxic drug levels, drug dosages will be lowered for two reasons:
- drugs that are typically highly protein bound will have more free drug circulating, and
- drugs that are highly lipophilic will have a larger volume of distribution.
Learning Activities
Clinical Reasoning and Decision Making Activities
1.Explain why seemingly small doses of morphine might have an unusually strong effect on someone with low serum albumin.
2. What if a drug is water soluble and highly protein bound? What will be its effect?
Note: Answers to the Clinical Reasoning activities can be found in the “Answer Key” sections at the end of the book.
References
Adams, M., Urban, C., El-Hussein, M., Osuji, J., & King, S. (2018). Pharmacology for nurses. A pathophysiological approach (2nd Canadian ed.). Pearson.
Burcham, J. R., & Rosenthal, L. (2019). Lehne’s pharmacology for nursing care (10th ed.). Elsevier.
Chippewa Valley Technical College, Egert, A., Lee, K., & Gill, M. (2023). Fundamentals of nursing pharmacology (1st Canadian ed.). BCcampus. https://opentextbc.ca/nursingpharmacology/chapter/1-9-examining-effect/
Fernandez, E., Perez, R., Hernandez, A., Tejada, P., Arteta, M., & Ramos, J. T. (2011). Factors and mechanisms for pharmacokinetic differences between pediatric population and adults. Pharmaceutics, 3(1), 53–72. https://doi.org/10.3390/pharmaceutics3010053
Le, J. (2024). Drug metabolism. Merck Manual. https://www.merckmanuals.com/en-ca/professional/clinical-pharmacology/pharmacokinetics/drug-metabolism
Onetto, A. J., & Sharif, S. (2023). Drug distribution. StatPearls. Retrieved October 20, 2025, from https://www.ncbi.nlm.nih.gov/books/NBK567736/
Media Attributions
- Figure 1.4a Blood brain barrier. (TRU Open Press).
- Figure 1.4b Blood brain barrier.png Creative Commons Attribution-Share Alike 4.0 International license. https://commons.wikimedia.org/wiki/File:Blood_brain_barrier.png
- Figure 1.4c Blood vessel with albumin molecules with bound and unbound drugs. (Sheila Odubote/ TRU Open Press) CC-BY-NC-SA
- Figure 1.4d Renault, Type r321, Bus by Emslichter, via Pixabay, is used under the Pixabay Content License.
- Figure 1.4.e Volume of distribution. (Sheila Odubote/ TRU Open Press).
A nearly impenetrable barricade that is built from a tightly woven mesh of capillaries cemented together to protect the brain from potentially dangerous substances such as poisons or viruses.
The rate at which 50% of a drug is eliminated from the bloodstream.
An unintended pharmacological effect that occurs when a medication is administered correctly.
The effects of a drug, other than the desired effect, sometimes in an organ other than the target organ.

